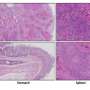
Discovery could lead to more effective PARP inhibitor drugs against cancer
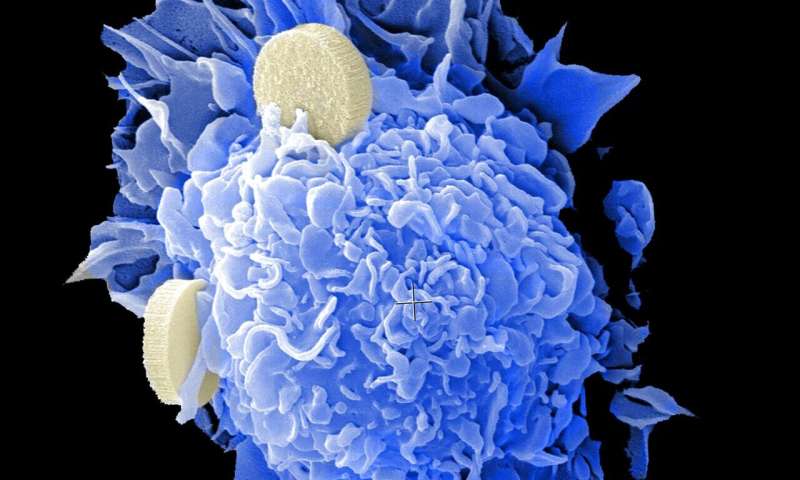
PARP inhibitors are recently developed drugs that have shown great promise against some types of ovarian, breast, prostate and pancreatic cancer, but many patients' tumors develop resistance to the drugs. Now researchers at the Perelman School of Medicine at the University of Pennsylvania have reported a strategy for defeating such resistance and boosting the effectiveness of these drugs.
The researchers, in a study published Nature Cell Biology, showed that an enzyme called ALC1 can powerfully reduce the effects of a PARP inhibitor by creating access for repair proteins to fix damaged subunits of DNA known as bases. This helps to keep treated tumors alive—whereas eliminating ALC1 activity removes this function and can exacerbate the type of damage that occurs when DNA repair deficient cells are treated with PARP inhibitors, thus dramatically increasing their effectiveness at destroying tumor cells.
"These findings suggest that PARP inhibitor drugs could be much more effective if used in combination with drugs that inhibit ALC1," said co-senior author Roger Greenberg MD, Ph.D., the J. Samuel Staub, MD, professor of Cancer Biology and director of the Penn Center for Genome Integrity.
The other co-senior author of the study was Junwei Shi, Ph.D., an assistant professor of Cancer Biology. The study's first author was Priyanka Verma, Ph.D., a postdoctoral researcher in the Greenberg Lab.
Since 2014 the United States Food and Drug Administration (FDA) has approved four PARP inhibitor drugs for the treatment of ovarian, breast, or related cancers that arise from mutations in BRCA1, BRCA2, and other DNA-repair genes. Inherited mutations to BRCA1 and BRCA2 are the major cause of hereditary breast and ovarian cancer. PARP (poly ADP ribose polymerase) is an enzyme that also helps with DNA repair. In cancer cells that already have lost key repair factors such as BRCA1 or BRCA2, PARP inhibitors deliver a further blow to DNA-repair capacity, ideally leading to unsustainable levels of DNA damage and either cell death or the cessation of growth.
In practice, many of these cancers either don't respond well to PARP inhibitor treatment, or evolve resistance after an initial response. Researchers have not fully understood how this happens.
In the study, Greenberg, Shi, Verma and their colleagues used a high throughput CRISPR gene-editing technology developed by Shi on BRCA1- and BRCA2-mutant cells to knock out other genes in the cells, to see if any of those gene disruptions altered the effectiveness of the PARP inhibitor drug olaparib. They found that knocking out the gene for ALC1, another DNA-repair-related enzyme, made the cancer cells dramatically more sensitive to olaparib, so that the drug killed the cancer cells at doses as much as 250 times lower than the usual lethal dose.
The researchers showed too that knocking out the ALC1 gene could defeat several known resistance mechanisms that sometimes evolve in BRCA-mutant cancers to permit their survival despite PARP inhibitor treatment.
The scientists obtained similar results in mouse models of BRCA-mutant cancers.
They note that ALC1's role in DNA-damage repair appears to be relatively dispensable when other DNA-repair mechanisms are intact, as they usually would be in non-cancerous cells. Thus, according to the researchers, adding ALC1 inhibitor drugs—though these have yet to be developed by pharma companies—may someday prove to be a good strategy for boosting the power of PARP inhibitors to help cancer patients.
More information: Priyanka Verma et al. ALC1 links chromatin accessibility to PARP inhibitor response in homologous recombination-deficient cells, Nature Cell Biology (2021). DOI: 10.1038/s41556-020-00624-3
Journal information: Nature Cell Biology