December 7, 2020 report
Photocatalytic nanofibers developed for use in efficient hydrogen production

A team of researchers from the U.K., Canada and Hong Kong has developed photocatalytic nanofibers that can be used for the efficient production of hydrogen. In their paper published in the journal Nature Chemistry, the group describes how their efficient nanofibers were made. Gregory Peterson, Sanghee Yang and Tae-Lim Choi with Seoul National University have published a News and Views piece in the same journal issue outlining the work done by the team
As scientists around the world continue to grapple with climate change, new ideas on ways to handle the challenge have emerged. One idea involves using hydrogen rather than gasoline to power vehicles and machines. Hydrogen is currently used in a wide variety of applications but its use has not become widespread due to issues such as cost of production and storage issues. In this new effort, the researchers focused on overcoming the problem of cost—their approach mimics nature by creating an artificial photosynthesis system capable of generating hydrogen from water with sunlight serving as the energy source.
In their work, the researchers began by noting that producing hydrogen using water could be made easier by carrying out just the conducting part of the reductive half-reaction for splitting water using a sacrificial reductant. They also noted that other attempts to make such systems have suffered from low quantum yield, poor stability and low catalyst turnover numbers. To get around these problems, the team developed a self-assembling nanofiber system.
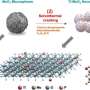
The system uses block copolymers and a crystallizable core-forming block along with a shell-forming block containing either a photosensitizer or a cobalt catalyst. This resulted in the creation of a self-assembly system where the materials grew into stable nanofibers with catalyst moieties and a photosensitizer in close proximity to one another. By distributing the components in gradient fashion, the nanofibers could be made into structures (with lengths from 95 to 3528 nm) that resembled bottle brushes without the internal wire. The resulting structure could then be used to convert water to hydrogen using sunlight.
More information: Jia Tian et al. Tailored self-assembled photocatalytic nanofibres for visible-light-driven hydrogen production, Nature Chemistry (2020). DOI: 10.1038/s41557-020-00580-3
Gregory I. Peterson et al. Polymers producing hydrogen, Nature Chemistry (2020). DOI: 10.1038/s41557-020-00582-1
Journal information: Nature Chemistry
© 2020 Science X Network