Accurate theoretical modeling unravels changes in molecules interacting with quantum light

A team of researchers from Italy, Norway, and Germany has demonstrated that the properties of molecules undergo significant changes when interacting with quantized electromagnetic fields in optical cavities. Using novel theoretical methodologies and computational simulations, the team revealed that the ground- and excited-state chemistry of molecules can be modified by a confinement in space. They show how the transfer of electrons inside the system can be controlled by modulating the frequency of the cavity field. Their newly-developed methodology could have a profound impact on many chemical and technological applications, such as photovoltaics, photochemistry, and optoelectronic devices. The team's work has now been published in Physical Review X and additionally highlighted in a Viewpoint by the journal.
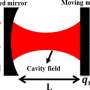
Cavity quantum optics deals with the interactions of photons and molecules inside an optical cavity, for example enclosed between two closely spaced mirrors. Perfect optical cavities can only support certain frequencies of light and they enhance the intensity of the associated electromagnetic field. This causes profound changes in the behavior of molecules which are placed into an optical cavity. In this situation, photons and molecules can couple and form new hybrid states known as polaritons. Importantly, these hybrid states exhibit properties of both the molecules and the photons. This means that their chemical behavior can be manipulated optically, for instance by tuning the photon energy and the cavity geometry. Hence cavities represent an entirely new lever for controlling molecular properties.
Yet the properties of molecules in cavities need to be better understood. While theoretical modeling in quantum optics offers an elaborate description of the electromagnetic field in the cavity, they provide an insufficient description of the molecule. So far, the only method that treats electrons and photons at the same level of quantization is quantum electrodynamical density functional theory, which is limited to situations where electrons and photons are uncorrelated.
However, the correlation between electrons and photons is crucial for capturing changes in molecular properties, even qualitatively. "These correlation effects were our focus," says Tor Haugland, Ph.D. student at the Norwegian University of Science and Technology and lead author of the paper. "Ours is the first ab initio theory that incorporates strong electron-photon correlation explicitly in a coherent and systematically improvable way."
The researchers extended the well-established coupled cluster theory for electronic structure to include quantum electrodynamics. Using this new framework, they showed that ground state potential energy surfaces are modified by the cavity close to conical interactions.
"This approach paves the way for novel strategies to control molecular chemistry," says co-author Enrico Ronca, a former postdoctoral researcher at the MPSD now based at the Institute for Physical-Chemical Processes of the National Research Council of Italy (IPCF-CNR). "We need solid theoretical methods in order to understand the fundamental processes which can helps us to manipulate atoms and molecules with quantum light."
The team's findings could significantly boost the current understanding of the relaxation pathways and photochemistry of molecules.
More information: Tor S. Haugland et al. Coupled Cluster Theory for Molecular Polaritons: Changing Ground and Excited States, Physical Review X (2020). DOI: 10.1103/PhysRevX.10.041043
Journal information: Physical Review X
Provided by Max Planck Society