The surprising strength of liquid crystals
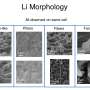
Dendrites are the destructive by-products of the cycle of charging and discharging lithium ion batteries. These tiny deposits form between the battery's anode and cathode, building up over time. Inevitably, they diminish battery life. More problematic is their risk of causing the battery to burst into flames. In the quest for safer and longer lasting batteries—especially for electric cars, trucks, and planes—researchers continue to explore methods to suppress the formation of dendrites.
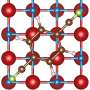
Researchers at Carnegie Mellon University have found that liquid crystals can be used as electrolytes with lithium metal anodes in batteries to suppress dendrite growth. Liquid crystals represent a new class of materials that have properties that are different from conventional liquids and solids. The dendrite suppression happens due to the tendency of liquid crystal molecules to line up in an ordered arrangement.
In findings published in the Proceedings of the National Academy of Sciences, the research team proposed various design criteria for selecting liquid crystals as battery electrolytes that can enable well-functioning lithium metal batteries.
"This comprehensive set of molecular level design rules will pave the way towards the realization of this new class of electrolytes for practical lithium metal batteries," said Venkat Viswanathan, associate professor of mechanical engineering at Carnegie Mellon.
"Batteries with increased energy density are critical to enable mass electrification of transportation. Lithium metal anodes offer a promising approach to improve the energy density of batteries, but dendrite formation plagues the safety and cycle life of lithium metal anodes," said Zeeshan Ahmad, the lead author on the paper.
The findings build on the research team's previous work on dendrite suppression using solid electrolytes. Although solid electrolytes provide superior dendrite suppression, they have slower lithium ion conductivities and cannot be integrated easily into the current lithium ion batteries. Liquids, on the other hand, have faster conductivity but cannot suppress dendrites.
The liquid crystal materials lie somewhere in between because they possess some orientational order but no positional order like solids. They are easily integrated into current lithium ion batteries, are safer, and offer spontaneous dendrite suppression.
Liquid crystals do have a disadvantage: their stability is not currently as good as that in current liquid electrolytes. The next step in this line of research is to further examine liquid crystalline materials so that they can satisfy all design criteria for future batteries.
More information: Zeeshan Ahmad et al. Design rules for liquid crystalline electrolytes for enabling dendrite-free lithium metal batteries, Proceedings of the National Academy of Sciences (2020). DOI: 10.1073/pnas.2008841117
Journal information: Proceedings of the National Academy of Sciences