Sensing carbon monoxide

Carbon monoxide is an insidiously toxic gas. It can pervade an enclosed space and causes drowsiness and at sufficiently high concentration is lethal to anyone breathing it. As such, there is a need for efficient and fast-reacting carbon monoxide sensor devices in a variety of industrial, commercial, and domestic settings. Devices are available but a new approach is discussed in the International Journal of Microstructure and Materials Properties that utilizes the chemistry of a twin film of molybdenum(VI) oxide and indium(III) oxide layers.
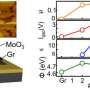
Physicists Nimba Kothawade and Arun Patil of the Arts, Science and Commerce College, and Vikas Deshmane of the SICES Degree College in Maharashtra, India, prepared thin films of MoO3:In2O3 using the spray pyrolysis technique on a glass substrate at 400 degrees Celsius. They characterized their film using X-ray diffraction, scanning electron microscopy, and energy-dispersive X-ray spectroscopy.
Once confident of their materials, the team then tested the electrical properties of their various formulations. They found that the resistivity of the films increased with MoO3 as the dopant in In2O3. They found a maximum resistivity of 1.75 × 104 Ωm for 0.3N (MoO3) and 0.1N (In2O3) binary oxide films.
They then tested the dual films gas-sensing characteristics against five different target gasses. The film composition ratio 0.3N:0.1N films had 70.50% sensitivity to 300 parts per million (ppm) of carbon monoxide gas at 150 degrees Celsius with high selectivity. The response time, the team reports was 15 seconds and recovery time was just 25 seconds.
More information: Nimba B. Kothawade et al. MoO3:In2O3 binary oxide thin films as CO gas sensor, International Journal of Microstructure and Materials Properties (2020). DOI: 10.1504/IJMMP.2020.110525
Provided by Inderscience