A new study unveils the mechanism of the nanoparticle gelation transition
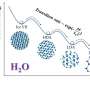
In a major breakthrough published in Nature Communications, the universal laws which govern the formation of nanostructured materials have been unveiled. Researchers led by Prof. Alessio Zaccone at University of Milan and by Prof. Peter Schall at University of Amsterdam, have demonstrated that the phase transition through which colloidal nanoparticles aggregate into a solid-like system-spanning material (a colloidal gel) is described by universal laws that are independent of the peculiar physico-chemical characteristics of a given system.
In particular, by means of a close synergy between theory, numerical simulations and experimental investigations, the researchers show, for the first time after decades of intense debate, that the underlying phase transformation (called colloidal gelation) coincides with a second-order continuous phase transition that occurs out of thermodynamic equilibrium. Phase transitions that lead, e.g., from a gas to a liquid or from a liquid to a solid are classified as first-order phase transitions if certain thermodynamic quantities present a discontinuity across the transition, whereas they are classified as second-order phase transitions if those thermodynamic quantities change smoothly.
This makes a huge difference, because the mathematical laws that allow predicting the transition point and its characteristics, as well as the physical properties of the new phase, are very different in the two cases. In the context of nanoparticles, the gelation transition is peculiar because the nanoparticles in the dispersed sol phase are suspended in a liquid (e.g., water) as single particles or part of "clusters" that are isolated from each other, whereas in the solid-like or gel phase the clusters interconnect into a fractal network. This network is apparently "disordered" or chaotic, but in reality, presents a high degree of symmetry because it is fractal. The fractal nature of the material implies that the density of particles decays in space with the same power-law as measured from every point in the material and the power-law exponent which governs this decay is called the fractal dimension (other examples of fractal objects are snow flakes, river networks, mountains or the coast of Britain).
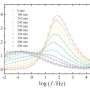
For decades, scientists have tried to determine whether the transformation of dissolved nanoparticles in a liquid into a fractal network is governed by a specific thermodynamic phase transition. The new study demonstrates that the phase transition, along with its critical exponents, which regulate the cluster size distributions both in the sol and in the gel phase, as well as the fractal dimension of the network itself (that is, the structure of the material), can be calculated theoretically a priori, and exactly the same values of exponents have been measured experimentally in colloidal systems using confocal microscopy techniques, and also the same exponents have been found in molecular dynamics simulations on the computer.
This result is a major step forward for the design, development and control of nanostructured materials with a desired fractal structure and to quantify and optimize the industrial synthesis of these materials. The applications are manifold and range from colloidal gels for agriculture (for the controlled release of active agents) to protein gels used in biotechnology and drug delivery, to nanocomposite rubber materials filled with nanoparticles fractal networks which allow for a reduction of polluting emissions in vehicle transport.
More information: Joep Rouwhorst et al. Nonequilibrium continuous phase transition in colloidal gelation with short-range attraction, Nature Communications (2020). DOI: 10.1038/s41467-020-17353-8
Journal information: Nature Communications
Provided by University of Milan