Researchers design a nanotechnology-based system that can transport methane at lower pressure and cost
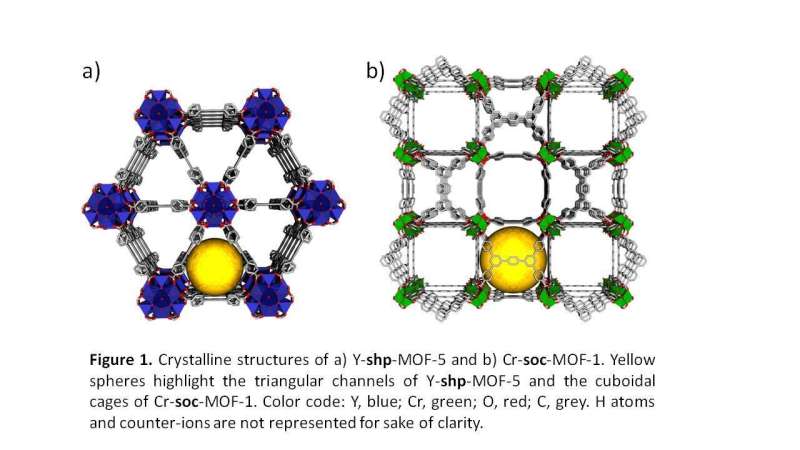
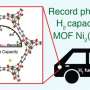
Researchers from the University of Alicante's Advanced Materials Laboratory have created an optimal, low-cost methane storage system. The team, led by UA Professor of Inorganic Chemistry Joaquín Silvestre, has used an MOF material (highly porous metal-organic framework). In the pores of this material, it is possible to create the conditions required to replicate gas structures under the sea, where millions of tonnes of natural gas are stored inside ice-like structures.
This system offers an alternative for natural gas transportation and natural gas-powered vehicles, like private cars, buses and ships. It is based on millimetric ice crystals that store gas, i.e. capture it and keep it stable.
The main advantage of these nanomaterials, Silvestre explains, is that with them "temperature can be reduced to 2°C, and pressure to about 60 bars." To liquefy natural gas and transport it by boat to different countries from its source, it must be at -162°C and at high pressures. Compressed gas-powered buses, conversely, require a pressure of 250 bars. With the system developed in this study, these two obstacles can be overcome, as the pressure and temperature conditions are more favourable.
This finding, published in the Journal of the American Chemical Society, results from the collaborative work between researchers from Saudi Arabia's King Abdullah University (KAUST), Morocco's Mohammed VI Polytechnic University, the U.S.'s Oak Ridge National Laboratory and the ALBA synchrotron radiation accelerator in Barcelona.
Background
MOF materials were first developed in the mid-1990s. So far, highly promising results have been obtained in areas as diverse as CO2 capture, methane storage, water treatment, sensors or biomedicine, among others.
More recently, studies led by researchers from Saudi Arabia have made it possible to synthesise new MOF materials with extremely high water adsorption capacity (up to 200 times their weight). "These hydrolytically stable materials have been proposed as systems that can capture water from the environment in arid areas, for instance during humid nights in coastal areas of the Mediterranean, which is later transformed into clean liquid water that can be drunk in the daytime," Joaquín Silvestre highlights.
In this sense, the UA researcher points out, "when I found out that my colleagues from Saudi Arabia and Morocco had designed perfect structures that could store that amount of water in their pores, I asked them for samples. They sent me two types of materials with, respectively, very small and slightly larger cavities. We checked that with the material having the smaller cavities the methane cannot form the crystal, but with the one with larger cavities it can."
Based on this, the UA-led study has been able to use the large amount of adsorbed water in the cavities of these MOF materials to create optimal methane storage systems. According to the UA expert, "by imitating nature, these MOF materials have been used as nanoreactors to encourage the growth of isolated methane hydrate crystals, in such a way that each pore of the parent structure contains a single crystal of hydrated gas."
Under this approach, the methane storage capacity of these materials could be increased by more than 50% compared to the same system on a dry basis. Most importantly, the load/unload pressure band is very small, which means that gas can be put into a container at a lower pressure and released by lowering such pressure.
With this reduction, in addition to its stability at a temperature of 2°C that is easily reached, costs are lower. Besides, the UA Professor adds that "nowadays, natural gas-powered vehicles require a very powerful and expensive compressor, which only some petrol stations can afford. However, by reducing pressure to 60 bars, a domestic compressor would be enough for storage purposes. We already have natural gas at home, so the car could be charged in the garage," he says.
Journal information: Journal of the American Chemical Society
Provided by Asociacion RUVID