Overcoming genomic imprinting barrier improves mammal cloning
Somatic cell nuclear transfer (SCNT, also known as cloning) technology holds great potential in animal production and regenerative medicine. However, the extremely low efficiency and frequently observed abnormalities in cloned embryos limit the development and application of this technology. Although the cloned embryos harbor complete genomic DNA sequence, multiple epigenetic barriers are the limiting factors of cloning efficiency.
Recently, researchers from the Institute of Zoology and the Institute of Genetics and Developmental Biology of the Chinese Academy of Sciences have collaborated and demonstrated experimentally that fixing the H3K27me3-mediated non-canonical imprinting can improve the cloning efficiency greatly.
The study, titled "Overcoming intrinsic H3K27me3 imprinting barriers improves post-implantation development after somatic cell nuclear transfer," is published online in Cell Stem Cell on June 18th, 2020.
Imprinting is a phenomenon that some genes only express one copy coming from either mother or father, but not the other copy.
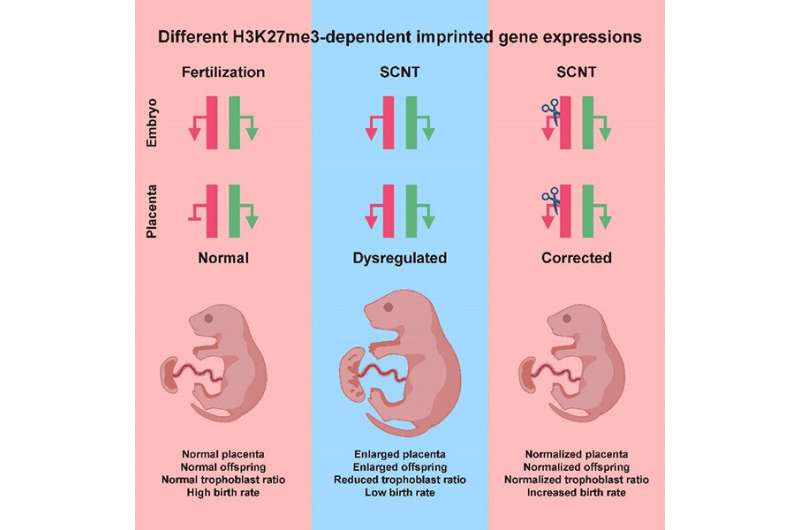
In this study, the researchers revealed that the H3K27me3-dependent imprinted genes were of aberrant biallelic expression in the post-implantation placenta of cloned embryos, and that is different from the monoallelic expression of these genes in normal embryos from fertilized eggs, suggesting that aberrant H3K27me3 imprinting may be an epigenetic barrier for animal cloning.
To demonstrate this experimentally, the researchers generated somatic cells with monoallelic deletion of four major H3K27me3-imprinted genes (Sfmbt2, Gab1, Smoc1 and Jade1) using haploid embryonic stem cell technology. They were first deleted in the haploid embryonic stem cell using CRISPR/Cas9-mediated gene editing.
Then these cells were used as artificial sperms to generate mice carrying monoallelic deletion of the four major H3K27me3-imprinted genes. The monoallelic expression of these genes can mimic the imprinted expression status of these genes.
Importantly, using somatic fibroblast cells from the aforementioned mouse, the researchers can achieve up to 14.2% (0 for wild-type fibroblasts) cloning efficiency. Meanwhile, the longstanding placenta and body overgrowth defects in cloned animals were also largely restored in these cloned mice.
Taken together, this study demonstrates that defect in H3K27me3-mediated imprinting is an important epigenetic barrier in mammalian cloning, and it establishes an elegant approach to overcome the barrier. The findings open a new avenue to improving the SCNT technology in multiple mammalian species.
More information: Le-Yun Wang et al. Overcoming Intrinsic H3K27me3 Imprinting Barriers Improves Post-implantation Development after Somatic Cell Nuclear Transfer, Cell Stem Cell (2020). DOI: 10.1016/j.stem.2020.05.014
Journal information: Cell Stem Cell
Provided by Chinese Academy of Sciences