April 2, 2020 report
Electrically neutral radical: A strong chemical reducing agent when exposed to light
A combined team of researchers from the University of North Carolina at Chapel Hill and Kent State University has discovered an electrically neutral radical that can be used as a strong chemical reducing agent when excited by light. In their paper published in the journal Nature, the group describes their process and possible uses for it. Radek Cibulka with the University of Chemistry and Technology, Prague, has published a News & Views piece describing the ways that light energy can be used to generate reactive molecules that undergo chemical transformations via methods that would ordinarily be difficult to achieve—and the work by the researchers in this new effort—in the same journal issue.
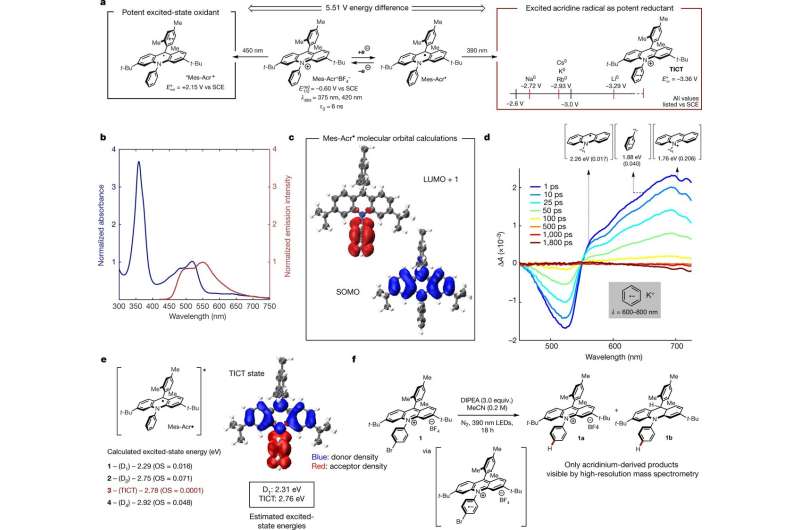
As Cibulka notes, prior research has shown that shining a light on certain molecules can force them to become more excited and thus more reactive than they would be in their natural ground state. Because of that, light has been used to produce reactive molecules that experience chemical transformations that would not normally occur—one example is oxidizing agents. More problematic has been using light to produce strong reductants. In this new effort, the researchers used light to generate molecules with reducing properties that are comparable to those produced using alkali metals.
The researchers note that photoinduced electron transfer (PET) reactions have typically been catalyzed using organic molecules that absorb light or transition-metal complexes such as chromium, ruthenium, copper or iridium—very few such reactions, however, have been conducted using neutral organic radicals as acceptors or excited-state donors. As part of their work investigating neutral organic radicals, they discovered one that had a maximum excited-state oxygen potential of −3.36 volts as compared to a saturated calomel electrode—a salt with a mesitylacridinium ion. When exposed to visible light, the species becomes an oxidant that steals an electron from a given substrate, resulting in conversion to an acridine radical.
The researchers suggest their discovery will likely lead to research by others into whether other molecules behave in similar ways—as well as renewed interest in using photocatalytic approaches in reductive systems.
More information: Ian A. MacKenzie et al. Discovery and characterization of an acridine radical photoreductant, Nature (2020). DOI: 10.1038/s41586-020-2131-1
Journal information: Nature
© 2020 Science X Network