January 23, 2020 feature
A design principle for creating selective and robust electrocatalytic interfaces
To effectively counteract climate change and meet rising global energy requirements, humans must drastically change their methods for generating energy. New catalysts for a carbon-neutral conversion of energy could be of great help in facing these challenges, facilitating the shift toward the use of renewable energy sources.
Electrolytic approaches, which convert electrical energy into chemical potential energy, are particularly promising for the development of catalysts to reduce CO2. These approaches are generally based on the use of aqueous substances that are cheap, readily available and safe for the environment. In addition, they typically work at ambient temperatures and pressures.
Some of the most common electrocatalysts used to enable CO2 reduction reactions are precious metals, base metals, metal oxides, metal dichalcogenides and molecular catalysts. These catalysts, as well as others tested in previous studies, often come with crucial limitations that prevent their large-scale implementation. For instance, they can be very expensive, while also exhibiting low energy efficiencies and unsatisfactory electrochemical stabilities.
Researchers at Stanford University have recently devised a new design strategy that could help to overcome some of these limitations, enabling the fabrication of selective and yet robust catalytic interfaces for heterogeneous electrocatalysts that could reduce CO2 to C2 oxygenates. Their approach for designing these electrocatalysts was introduced and outlined in a paper published by Nature Energy.
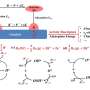
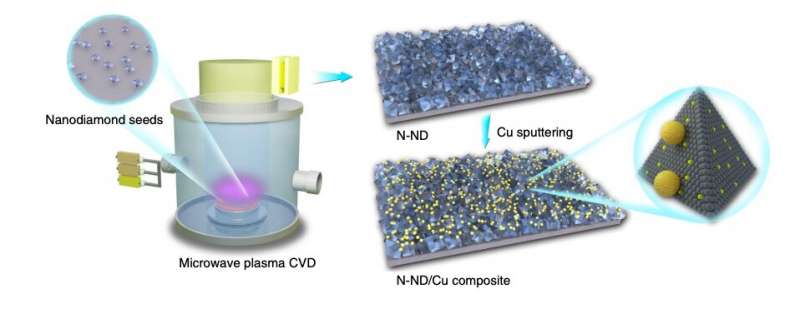
"We report a design principle for the creation of a selective yet robust catalytic interface for heterogeneous electrocatalysts in the reduction of CO2 to C2 oxygenates, demonstrated by rational tuning of an assembly of nitrogen-doped nanodiamonds (N-ND) and copper nanoparticles," the researchers wrote in their paper.
In their study, the team at Stanford essentially showed how a catalytic interface can be assembled by incorporating Cu nanoparticles into N-ND, creating the material N-ND/Cu. The synergy of these two merged components (i.e., Cu and N-ND) was found to enable significant improvements in the catalysts' process of CO2 to C2 oxygenate transformation.
"The catalyst exhibits a Faraidic efficiency of -63 percent toward C2 oxygenates at applied potentials of only -0.5V versus a reversible hydrogen electrode," the researchers wrote in their paper. "Moreover, this catalyst shows an unprecedented persistent catalytic performance up to 120h, with steady current and only 19 percent activity decay."
The catalyst developed using the design principle proposed by these Stanford-based researchers was found to outperform existing electrocatalytic systems in several domains, achieving a remarkably high activity and selectivity. In addition, the new design strategy allows for an unparalleled degree of control over the catalytic interface, and consequently also over the reaction energetics and kinetics.
In the future, the approach could guide the development of a variety of new electrocatalytic interfaces, paving the way for more effective and environmentally friendly techniques for storing energy. In addition, the same design strategy should be easy to apply to the fabrication of numerous catalytic transformations, particularly those based on the use of renewable energy sources and readily available aqueous substances.
More information: Hongxia Wang et al. Synergistic enhancement of electrocatalytic CO2 reduction to C2 oxygenates at nitrogen-doped nanodiamonds/Cu interface, Nature Nanotechnology (2020). DOI: 10.1038/s41565-019-0603-y
Journal information: Nature Nanotechnology , Nature Energy
© 2020 Science X Network