A new antiseizure target?

Temporal lobe epilepsy (TLE) is the most common form of epilepsy with focal seizures, and about one-third of individuals with TLE experience seizures that do not respond to medical treatment.
To search for new drug targets, Dane Chetkovich, MD, Ph.D., and colleagues explored the regulation of ion channels called "HCN channels," which have been linked to aberrant neuronal activity in TLE.
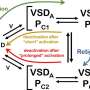
They focused on a protein called TRIP8b that regulates the subcellular location and function of HCN channels. The investigators demonstrated that TRIP8b phosphorylation (the addition of a phosphate chemical group to a certain site on the protein) enhances binding to HCN channels and influences channel electrical activity. They also showed that TRIP8b phosphorylation is reduced in an animal model of TLE.
The findings, reported in the Journal of Biological Chemistry, suggest that loss of TRIP8b phosphorylation may affect HCN channel properties and contribute to the development of epilepsy. Drugs that restore TRIP8b phosphorylation might enhance HCN function and reduce neuronal excitability in epilepsy.
More information: Kendall M. Foote et al. Phosphorylation of the HCN channel auxiliary subunit TRIP8b is altered in an animal model of temporal lobe epilepsy and modulates channel function, Journal of Biological Chemistry (2019). DOI: 10.1074/jbc.RA119.010027
Journal information: Journal of Biological Chemistry
Provided by Vanderbilt University