A new material for regenerative medicine capable of controlling cell immune response
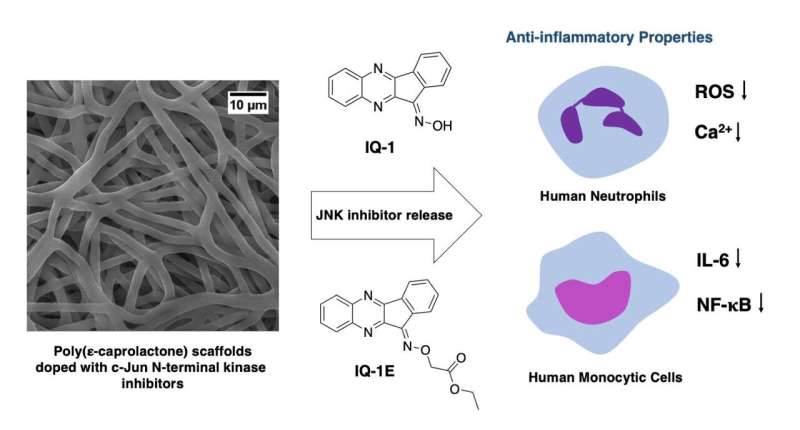
Scientists at Tomsk Polytechnic University jointly with the University of Montana (USA) proposed a promising new material for regenerative medicine for recovery of damaged tissues and blood vessels. This is a 3-D scaffold, made of biodegradable material and filled with special inhibitors, which were also obtained at TPU. They literally turn off the work of enzymes, responsible for the inflammation reaction, occurring in immune cells in response to external stimuli. In this case, such an irritant is a regenerative material.
According to scientists, the proposed solution is a simpler way to control the immune response compared to existing ones. The results were published in ACS Biomaterials Science & Engineering.
"Nowadays, researchers have only a few tools to regulate the immune response. You can work with proteins, but it is difficult. You can use compounds, capable of killing immune cells, but they are harmful to other cells.
We followed a different path and suggest using inhibitors placed directly in the material itself to recover damage," said Ksenia Stankevich, author of the article and engineer at the Laboratory for Plasma Hybrid Systems.
Scaffolds are 3-D frames of thin polymer fibers interwoven with each other in different directions. In regenerative medicine, they are used in case of injuries of bone and soft tissues. They are placed in the damaged area and new tissue regenerates through the scaffold and fills the injured area.
TPU and the University of Montana used a biodegradable polycaprolactone polymer for their scaffolds. It makes products more flexible and affordable in comparison with alternatives. The scaffolds made of polycaprolactone were created using the method of electrospinning, producing the thinnest fibers from a polymer solution under the electric field. At the stage of obtaining the scaffolds, we introduce inhibitors into the polymer structure. These are two compounds—IQ-1 (full name—11H-indeno [1,2-b] quinoxaline-11-on oxime) and IQ-1E (full name—11H-indeno [1,2-b] quinoxaline-11- on O-(O-ethylcarboxymethyl) oxime).

"Inhibitors suppress or slow down physiological and physicochemical processes. They affect enzymes. To do this, the enzyme and the inhibitor must fit together like a lock and a key. One of the groups of enzymes responsible for the inflammatory process is the JNK group," Ksenia Stankevich explains.
"Earlier we obtained promising new inhibitors, demonstrating high biological activity in inhibiting the functioning of these enzymes, such as IQ-1 and IQ-1E. Our scaffolds differ in the use of specific inhibitors and also in the fact that we can release them from the material gradually, having a prolonged effect. This is mainly due to the gradual natural degradation of the polymer. Additionally, it degrades to biocompatible 6-hydroxycaproic acid, which is recycled by human body."
The immune response of a cell is a cascade of biochemical processes. In this case, the JNK enzymes are links in the chain. Inhibitors bind to enzymes and block their work. Thus, in suppressing one link, we turn off the entire subsequent reaction chain.
"In this article, we present the research results on immune cells, isolated from human blood and cell lines. In the future, we will look for opportunities for in vivo research. Eventually, our scaffolds could be used to recover damages of soft tissues and blood vessels. The polycaprolactone has all suitable mechanical properties. For instance, it can reduce the negative consequences after a heart attack and stroke," the researcher says.
"Scaffolds from various materials are already being implemented into medical practice in developed countries, but it is too early to talk about their widespread application. However, it is only a matter of time—that is why scientists continue searching for the most effective materials and biologically active compounds. "
More information: Ksenia S. Stankevich et al, Poly(ε-caprolactone) Scaffolds Doped with c-Jun N-terminal Kinase Inhibitors Modulate Phagocyte Activation, ACS Biomaterials Science & Engineering (2019). DOI: 10.1021/acsbiomaterials.9b01401
Provided by Tomsk Polytechnic University



















