Solvent pH controls interactions of gold nanoclusters, offers potential applications in drugs and imaging

The properties of gold in nanoscale are significantly different to those of bulk gold. Of special interest are gold nanoclusters, that are composed of between tens to some hundreds of gold atoms. Numerous of such cluster structures are known and synthesizable to atomic precision. The aim of this thesis was to apply molecular dynamics simulations on investigating properties of gold nanoclusters in different environments. The simulations reveal that gold nanocluster can bind to viruses by different interactions, and that the strength of the interactions are dependent on pH conditions.
The applicability of gold nanoclusters in medicine is widely studied. At the University of Jyväskylä, their usage has been demonstrated for example in virus imaging. Gold nanoclusters are generally composed of a gold core covered by a protective layer of different molecules. The protecting layer thus essentially determines how the gold nanocluster interacts with its environment. Moreover, the properties of the gold nanoclusters may be altered by tailoring the type of the molecules in the protective layer.
The aim of M.Sc Emmi Pohjolainen's dissertation at University of Jyväskylä, Finland, was to study different gold nanoclusters in various environments by means of molecular dynamics simulations. Molecular dynamics simulations are an established tool in studies of systems whose properties and dynamics need to be investigated in atomic precision, while keeping the computation time reasonable.
While molecular dynamics simulations have been widely used in studies of biomolecules, their use in metal nanocluster research has been relatively sparse. The very first aim of this thesis was to develop and validate parameters to enable simulating such systems. These parameters have since been used by other groups outside the University of Jyväskylä as well.
The acidity controls gold nanoclusters' binding to virus
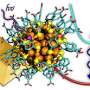
All the simulation results essentially need to be connected to the experimental data. On one hand the experimental results may be complemented by simulation results, on the other hand available experimental information need to be used in validating the goodness of the simulation. The simulations performed for this thesis included for example simulating gold nanoclusters in interactions with virus, by constructing a system with a full virus capsid covered with 60 gold nanoclusters. This system contained some 3.5 million atoms, and is as such a notably large system to simulate at atomistic scale.
The results revealed that gold nanoclusters may interact with the virus by different means, and the strength of these interactions is dependent on the pH conditions. This information may be in future utilized in design of imaging and drug molecules that need to bound to specific locations on the virus surface. Also binding of different types of drug molecules to the virus were simulated, and binding strengths were compared to those of gold nanoclusters.
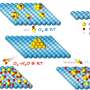
In this thesis also self-assembly of gold nanoclusters into flakes or spherical structures, previously observed experimentally, were simulated. The simulations revealed that the stability of such superstructures is dependent on both solvent conditions and the distribution of charges on the cluster surface. Thus, self-assembling or disassembling could be controlled by changing the solvent and pH conditions. This property could be usable for example in drug-carrier molecules.
More information: M.Sc. Emmi Pohjolainen. Atomistic Simulation View on Gold Nanocluster Functionalities via Ligand Shell Dynamics. jyx.jyu.fi/handle/123456789/64255
Provided by University of Jyväskylä