Resonance-enhanced tunneling induces flourine and para-hydrogen reaction in interstellar clouds

Scientists from the Dalian Institute of Chemical Physics (DICP) of the Chinese Academy of Sciences and their collaborators investigated the mechanism of rapid reactivity of the F + H2 reaction at low temperature and found that rapid reactivity was actually induced by resonance-enhanced tunneling.
This finding explains the observation of HF in interstellar clouds, which is generated only through the F + H2 reaction. The research was published in Nature Chemistry.
Generally, a chemical reaction with an energy barrier can only happen at collision energies higher than the barrier. However, quantum tunneling at energies below the reaction barrier plays a significant role in many chemical processes, especially at low temperature.
Chemical reaction plays an important role in the evolution of interstellar clouds. In interstellar space, temperature is particularly low, thus quantum effects in reactions may play a significant role.
HF in interstellar clouds was first discovered in 1997, and recent observations have found that HF is ubiquitous in the universe. Since the F + H2 reaction, with an energy barrier of 1.8kcal/mol, is the sole source of observed HF at low temperature in interstellar clouds, how does it rapidly proceed? Even considering normal quantum tunneling, the reaction rate is too low to be observed with a reaction barrier of such height (~800K).
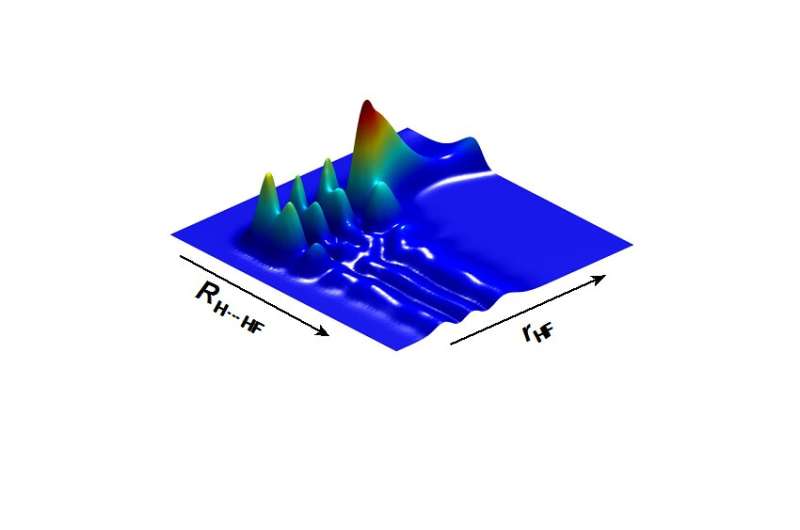
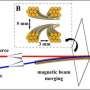
With improved molecular crossed beam apparatus, the scientists measured the quantum state specific backward scattering spectroscopy (QSSBSS) as a function of collision energy in the range 1 ~ 35 meV. A peak in QSSBSS was clearly observed at about 5 meV. Using detailed dynamics analysis on an accurate potential energy surfaces (PESs), they found that the peak was produced by the ground resonance state of the F + H2 to HF + H reaction. They also discovered that the oscillations at about 20 meV were produced by the first excited resonance state of the F + H2 reaction.
Further theoretical analysis indicated that if the contribution of the resonance-enhanced tunneling were removed from the reactivity, the reaction rate constant of F + H2 below 10K would be reduced more than three orders of magnitude.
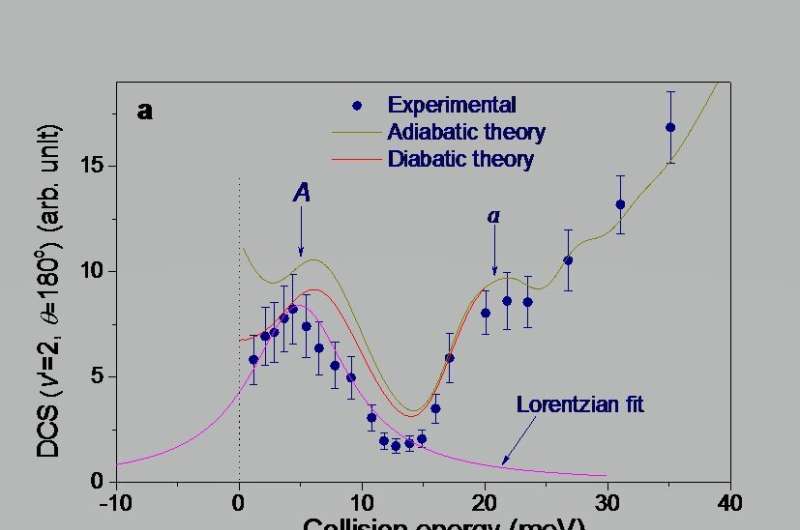
Thus, the reactivity of the F + H2 reaction is almost completely derived from resonance-enhanced tunneling from the ground resonance state. With an accurate PES, the theory provides the reaction rate constant for the F + H2 reaction over a wide temperature range, which is essential to understanding interstellar chemistry.
More information: Enhanced reactivity of flourine with para-hydrogen in cold interstellar clouds by resonance-induced quantum tunnelling, Nature Chemistry (2019). DOI: 10.1038/s41557-019-0280-3 , www.nature.com/articles/s41557-019-0280-3
Journal information: Nature Chemistry
Provided by Chinese Academy of Sciences