May 17, 2019 report
A more efficient way to cleave pendant olefins from terpenes and similar compounds
A team of chemists at the University of California has found a more efficient way to separate olefins from terpenes and similar compounds. In their paper published in the journal Science, the group describes the new approach and possible applications for its use. Seb Caille with Amgen has published a Perspective piece outlining the work by the team at UC in the same journal issue and explains why it is important.
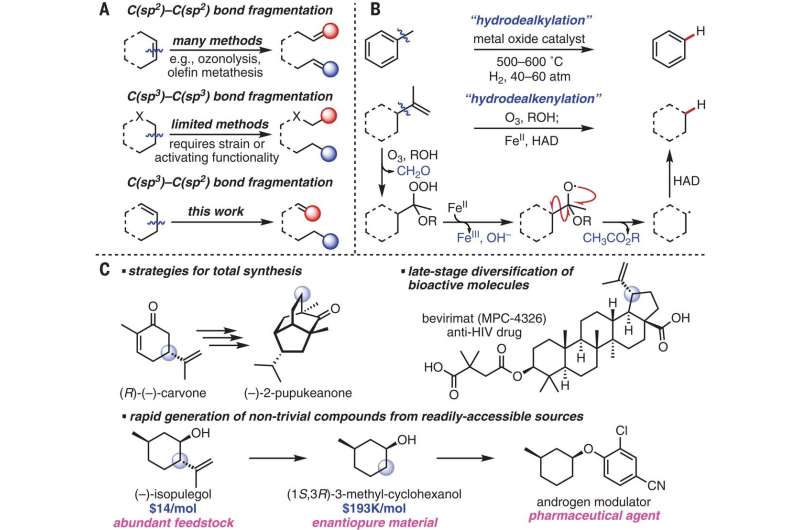
As medical science reveals more about the ways our bodies work, pharmaceutical companies continuously work to develop therapies that make use of new findings. But as Caille explains, such work is often beleaguered by problems associated with the study of ever more complex molecular structures. Many times, when a particular molecule or structure is found to be useful, it must first be extracted from its more complex state. In practical terms, this can mean putting a material through several steps as part of a manufacturing process. Caille also notes that generally, the more steps that are required to process a material, the more it costs to produce an end result, such as a drug. For this reason, chemists work to find ways to achieve the same ends using fewer steps. In this new effort, the researchers have developed a new way to separate pendant olefins from terpenes.
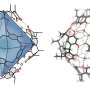
Pendant olefins are a type of open-chain hydrocarbon with at least one bond. And terpenes, another kind of hydrocarbon, are volatile unsaturated hydrocarbons that are often found in the natural oils produced by plants, particularly trees. Pharmaceutical companies have found that such olefins can be useful in a wide range of applications, but it takes many steps to separate them from the hydrocarbons in which they are found—this is because of their unique carbon framework.
The new process by the team at UC involves successively treating cooled terpenes with ozone and providing a hydrogen atom donor in a nonanhydrous solvent. They note that the treatment works because ozone is an iron oxidant. They suggest their single-process treatment could replace multiple steps used in other processing systems.
More information: Andrew J. Smaligo et al. Hydrodealkenylative C(sp3)–C(sp2) bond fragmentation, Science (2019). DOI: 10.1126/science.aaw4212
Journal information: Science
© 2019 Science X Network