Carbon dioxide mineralization in geologically common rocks for carbon storage
Humanity needs to improve when it comes to reducing carbon emissions to prevent the worst effects of climate change. If the world is to meet the IPCC's minimum target of keeping global temperature increases below 1.5 °C, every possible avenue for CO2 remediation must be explored.
Geological trapping can play a major role here. Our planet's underground rocks and sediments offer a vast potential space for long-term carbon storage. To support this, a recent computational study from a Japanese-led international group at Kyushu University shows how trapped carbon dioxide can be converted into harmless minerals.
The rocks beneath the earth's surface are highly porous, and trapping involves injecting CO2 into the pores after collecting it from its emission source. Although CO2 is usually considered too stable to react chemically with rock, it can bind tightly to the surface by physical adsorption. Eventually it dissolves in water, forming carbonic acid, which can react with aqueous metals to form carbonate minerals.
"Mineralization is the most stable method of long-term CO2 storage, locking CO2 into a completely secure form that can't be re-emitted," explains Jihui Jia of the International Institute for Carbon-Neutral Energy Research (I2CNER), Kyushu University, first author of the study. "This was once thought to take thousands of years, but that view is rapidly changing. The chemical reactions are not fully understood because they're so hard to reproduce in the lab. This is where modeling comes in."
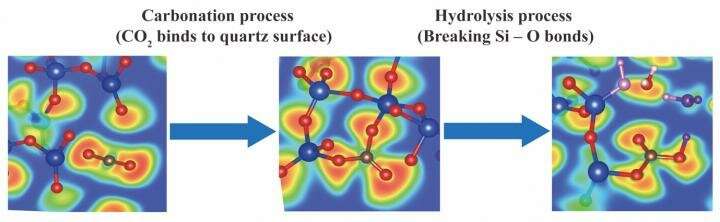
As reported in The Journal of Physical Chemistry C, simulations were initially run to predict what happens when carbon dioxide collides with a cleaved quartz surface—quartz (SiO2) being abundant in the earth's crust. When the simulation trajectories were played back, the CO2 molecules were seen bending from their linear O=C=O shape to form trigonal CO3 units bonded with the quartz.
In a second round of simulations, H2O molecules were added to mimic the "formation water" that is often present beneath oil and gas drilling sites. Intriguingly, the H2O molecules spontaneously attacked the reactive CO3 structures, breaking the Si-O bonds to produce carbonate ions. Just like carbonic acid, carbonate ions can react with dissolved metal cations (such as Mg2+, Ca2+, and Fe2+) to bind carbon permanently into mineral form.
Together, the simulations show that both steps of CO2 mineralization—carbonation (binding to rock) and hydrolysis (reacting with water)—are favorable. Moreover, free carbonate ions can be made by hydrolysis, not just by dissociation of carbonic acid as was once assumed. These insights relied on a sophisticated form of molecular dynamics that models not just the physical collisions between atoms, but electron transfer, the essence of chemistry.
"Our results suggest some ways to improve geological trapping," says study lead author Takeshi Tsuji. "For quartz to capture CO2, it must be a cleaved surface, so the silicon and oxygen atoms have reactive 'dangling' bonds. In real life, however, the surface might be protected by hydrogen bonding and cations, which would prevent mineralization. We need a way to strip off those cations or dehydrogenate the surface."
Evidence is growing that captured CO2 can mineralize much faster than previously believed. While this is exciting, the Kyushu paper underlines how complex and delicate the chemistry can be. For now, the group recommends further studies on other abundant rocks, like basalt, to map out the role that geochemical trapping can play in the greatest technical challenge facing civilization.
More information: Jihui Jia et al, Ab Initio Molecular Dynamics Study of Carbonation and Hydrolysis Reactions on Cleaved Quartz (001) Surface, The Journal of Physical Chemistry C (2019). DOI: 10.1021/acs.jpcc.8b12089
Journal information: Journal of Physical Chemistry C
Provided by Kyushu University