Do alpha particle condensates exist in oxygen nuclei?
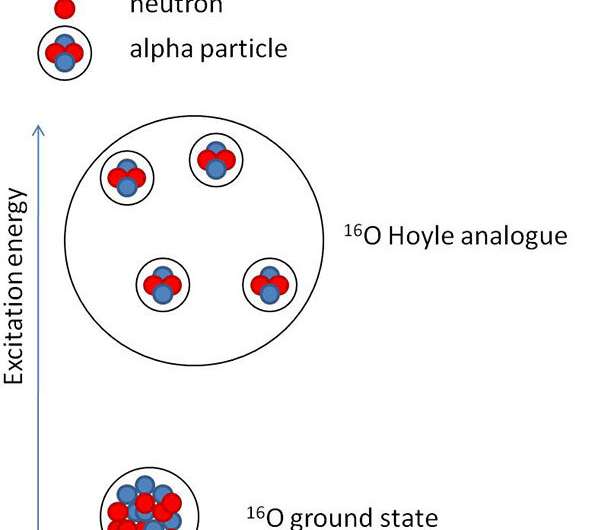
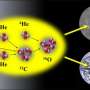
Nuclei in their lowest energy states (ground state) are composed of neutrons and protons. Two protons and two neutrons in a nucleus can cluster together to form alpha particles. When the nucleus gets nearly enough energy to disintegrate into alpha particles, the alpha particles can arrange themselves in the lowest possible quantum energy level, forming a Bose-Einstein condensate. Examples are the ground state of beryllium-8 and the famous carbon-12 "Hoyle" state, named for Fred Hoyle who first postulated its existence to explain the production of carbon in stars. Could analogous states exist in other isotopes like oxygen-16 and neon-20? Nuclear researchers at Texas A&M University indicated a state analogous to the Hoyle state exists in oxygen-16.
The existence of the Hoyle state in carbon-12 is very important. In fact, it's thanks to this state that carbon-12, the key element for life as we know it, could be formed in the early universe. The carbon-12 Hoyle state also has peculiar characteristics. These features can be explained by describing carbon as a diluted gas of alpha particles, implying the existence of a new state of nuclear matter analogous to the well-known Bose-Einstein condensate for molecules. Finding states analogous to the carbon-12 Hoyle state in heavier nuclei will show that the Hoyle state is not a lucky occurrence in carbon-12. Rather, it's a state of nuclear matter that can be found in other nuclei under similar conditions.
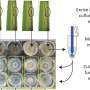
The identification and the study of states analogous to the Hoyle state in heavier nuclei can provide a test for the existence of alpha condensates in nuclear matter. At the Cyclotron Institute of Texas A&M University, researchers studied the reaction between neon-20 and alpha particles using a thick helium target and a neon-20 beam. The team adjusted the pressure of the helium gas to stop the beam before the detectors placed at the end of the experimental chamber. As the beam travels in the chamber, it gradually loses energy so that systems of different excitation energy can be formed at different positions inside the gas. The team detected events producing one, two, three, and up to four alpha particles during the experiment. The detectors placed at the end of the chamber measured the energies and the positions of the incoming particles as well as distinguished alpha particles from other reaction products. The analysis of the data from events that produced three alpha particles allowed the team to identify the Hoyle state in carbon-12. The decay of that state into three alpha particles agreed with other data in the literature. Although the statistics on events with four alpha particles were low, the team could identify a structure at about 15.2 MeV that could correspond to a state analogous to the Hoyle state in oxygen-16. Previously, researchers observed this state, but they didn't observe its decay into four alpha particles, confirming the alpha-cluster nature of this state. Further analysis of the decay path shows that the decay into four alpha particles proceeds with equal probability through emission of two beryllium-8 in the ground states or through the emission of an alpha particle and a carbon-12 in the Hoyle state.
More information: M. Barbui et al. Searching for states analogous to the C12 Hoyle state in heavier nuclei using the thick target inverse kinematics technique, Physical Review C (2018). DOI: 10.1103/PhysRevC.98.044601
Provided by US Department of Energy