Tumor cells conquer territory from their neighbors using a newly discovered mechanism
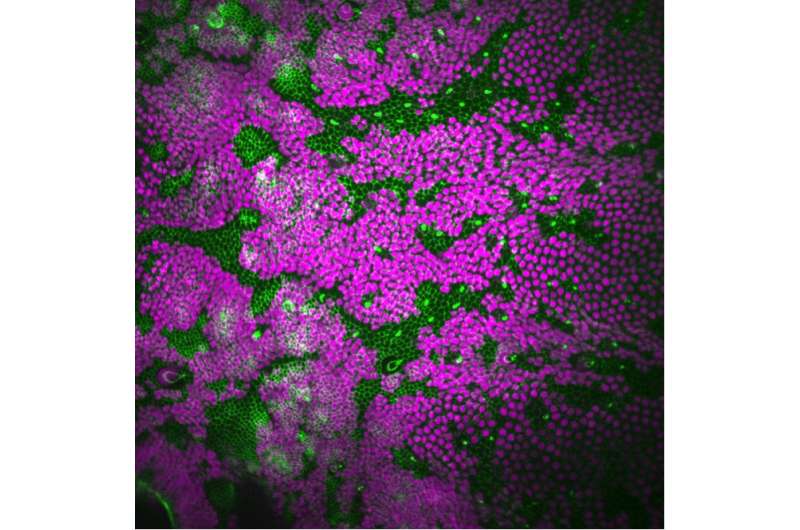
Despite decades of cancer research, the early phases of tumour progression that connect the appearance of few abnormal cells to the formation of a clinically detectable tumour mass remains poorly understood. It was previously proposed that certain mutations could give a competitive advantage to a subset of cells that would enable them to kill and replace their neighbours, thereby initiating a cancerous tumour. Yet, the mechanisms surrounding the basis of such competition were not clear. Researchers from the Institut Pasteur and the Champalimaud Centre for the Unknown, have just discovered a new mechanism that may explain how tumoural cells can eliminate their neighbours and spread throughout the body.
In a study published two years ago, Eduardo Moreno from the Champalimaud Centre for the Unknown and Romain Levayer from the Institut Pasteur, identified a form of competition between cells that was not previously known, which they named mechanical competition. "During normal development, for example, there are phases in which tissues become overcrowded and then some cells are eliminated," says Moreno. "But it was thought that these cells were being pushed out of the overcrowded tissue by live cell extrusion, meaning they would kind of just pop out."
The team discovered that this was not the case. Cells were not extruded alive from the tissue. Instead, they were actively killed by a form of competition that was previously unknown. "When we blocked the programmed cell death pathway, these cells would actually compress and compress, but they wouldn't die, nor get extruded. That's when we realised that there must be a different type of competition, a mechanical one, in which cells somehow sense the increasing physical pressure and use it to eliminate their neighbours," Moreno and Levayer explain.
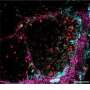
In this new study, published December 13th, 2018 in the scientific journal Current Biology, the researchers went one step further and identified the molecular mechanism that leads the compressed cells to be eliminated. For that, they chose to focus on epithelial bodily tissues, such as those that form the skin and the digestive system. "Epithelium is the most common tissue in our body. It is composed of layers of cells that form barriers separating the inside from the outside," Levayer explains. "The majority of human tumours (roughly 90 percent) arise from epithelium."
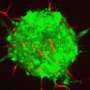
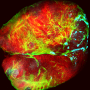
Using an epithelium of the fruit fly Drosophila melanogaster as a model system and a combination of tools from biology and physics, the researchers found that an internal cellular pathway called EGFR/ERK, a well-known regulator of cell survival, was modulated by mechanical stress.
Specifically, they found that when healthy cells were compressed by tumoural cells, first the survival-promoting EGFR/ERK signal in the healthy cells decreased, and then they died. Importantly, when the same pathway was artificially activated in compressed healthy cells, their elimination was prevented and the expansion of the tumoural cells was slowed-down.
Why do the tumour cells win against the healthy cells in this mechanical competition? It could go either way in principle, since they are both subjected to the same external forces. Moreno explains that the tumour often has the advantage of having their self-elimination pathways blocked. "It's one of the hallmarks of cancer," he points out. "In addition to them being more proliferative, they also have all these apoptotic (self-elimination) pathways mutated, so they are more resistant to death."
Together, these results make the EGFR/ERK pathway a prime candidate as an important cellular mechanism through which mechanical competition works and suggest a possible role for it in future clinical applications.
"The identification of the pathway that senses deformations and triggers cell elimination is an important step forward. It suggests that preventing the elimination of healthy cells that surround tumours by preventing the decreased activation of this pathway, may be a new therapeutic strategy to halt tumour growth and reduce cancer morbidity in the future," concludes Levayer. Future work will help to find how general is this mechanism and whether it is conserved in mammals. This observation may also have consequences for the understanding of how our tissues are formed during embryogenesis and how their size and shape are regulated.
More information: Eduardo Moreno, Florence Levillayer, Léo Valon and Romain Levayer, Competition for space induces cell elimination through compaction-driven ERK downregulation, Current Biology (2018). DOI: 10.1016/j.cub.2018.11.007
Journal information: Current Biology
Provided by Champalimaud Centre for the Unknown