December 6, 2018 report
Researchers propose a new theory to explain iron-oxide concretions found in Utah and Mongolia

A team of researchers from Japan, Mongolia and the U.K. has developed a new theory to explain the origin of iron-oxide concretions (hard, solid masses) found in Utah and Mongolia. In their paper published in the journal Science Advances, the group describes their theory and how well it tested.
People have known about the iron-oxide balls found in Utah for hundreds, if not thousands of years—they are often referred to as Moqui marbles after the local Indian tribe. But how the small spheres came to exist is a mystery. It is the same story with the little balls in Mongolia. Interest in both has spiked in recent years due to the "blueberries" (hematite spherules) found on Mars back in 2004—many geologists believe they developed in the same way as the balls on Earth. In this new effort, the researchers suggest they may have solved the mystery of their origin on Earth, and by extension, on Mars, as well.
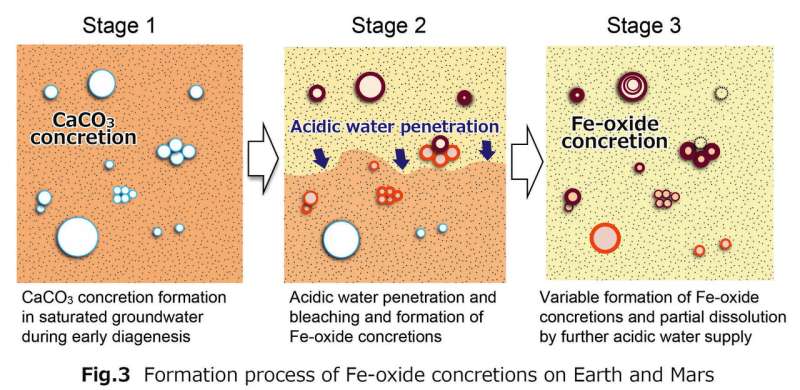
The researchers started by looking at samples of the balls using X-ray diffraction and optical microscopes. This allowed them to get a good look at the cores of the balls. After studying the makeup of the interior of the balls, the researchers concluded that they were first nothing but sandstone (mostly calcite). But then, the sandstone was very slowly dissolved by acidic water rich in iron, resulting in the formation of the balls, which notably have calcite cores. The researchers next conducted lab experiments designed to make similar balls using the same process. They report that their efforts were successful.
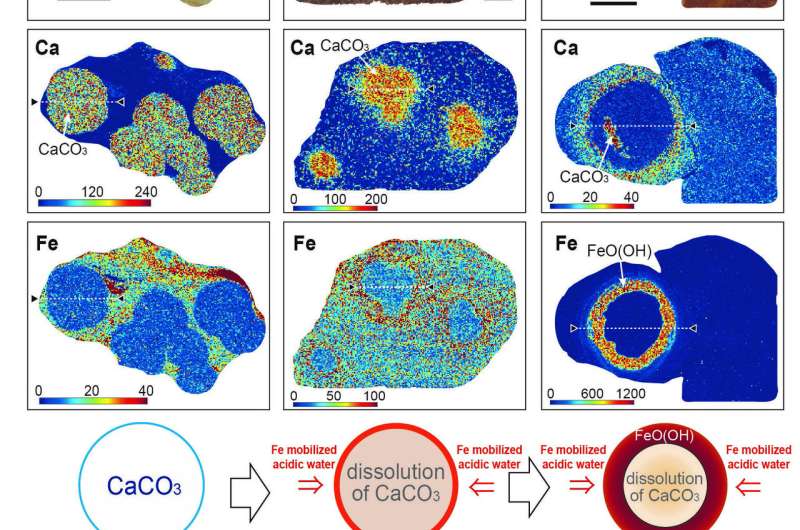
The researchers suggest their findings offer evidence of flowing water on Mars during the time the "blueberries" came to exist. They note the lack of a calcite core does not mean it was never there—such cores could very easily have vanished due to over-wash flowing through the rock. They further suggest that such an occurrence would also explain the dearth of carbonate on Mars' surface—it would have been dissolved as part of the process that led to the creation of the blueberries.
More information: H. Yoshida et al. Fe-oxide concretions formed by interacting carbonate and acidic waters on Earth and Mars, Science Advances (2018). DOI: 10.1126/sciadv.aau0872
Journal information: Science Advances
© 2018 Science X Network