Seeing a salt solution's structure supports one hypothesis about how minerals form
Scientists at the Department of Energy's Oak Ridge National Laboratory used neutrons, isotopes and simulations to "see" the atomic structure of a saturated solution and found evidence supporting one of two competing hypotheses about how ions come together to form minerals.
The study, reported in The Journal of Physical Chemistry B, may improve understanding of ion–water interactions in catalysis, environmental remediation and industrial engineering.
"The precise measurement we made has implications for all kinds of mineral formation reactions and waste issues in subsurface geologic environments, such as those containing nuclear wastes or hydraulic fracturing fluids," said ORNL geochemist Hsiu-Wen Wang. "Bringing together intense neutrons at the Spallation Neutron Source and our advanced computational models allowed us to do this measurement, which was not possible before."
Isotopes played a key role, too. Because different isotopes of an element contain the same number of protons but varying numbers of neutrons, a neutron beam hitting one isotope will scatter neutrons slightly differently than it would striking another isotope.
DOE prioritizes geochemistry research because about 80 percent of our energy comes out of the ground, through which fresh water or salt water permeates. The structure and dynamics of these aqueous solutions affect chemical reactions, molecular conformations, and mineral formation and dissolution.
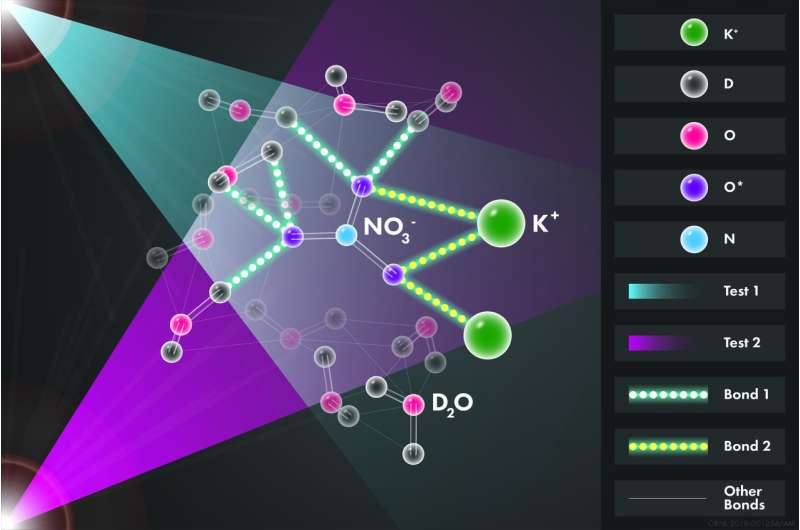
"One hypothesis is that ion pairs come together to form minerals, and a competing idea is that an extended network of ions exists in these solutions," said Andrew Stack, who leads ORNL's Geochemistry and Interfacial Sciences group. "We found ion pairs in this case but did not find a network."
In a glovebox that reduces exposure to humidity, Wang and Stack made two ultrapure solutions of KNO3 dissolved in D2O, or "heavy water." In D2O, the isotope deuterium (D) replaces hydrogen (H) in water's chemical formula. Deuterium reduces background noise in the experiment. The only difference between the two salty solutions was which oxygen (O) isotope labeled molecules of nitrate (NO3-)—either naturally predominant 16O or rarer 18O. A positively charged ion of potassium (K+) served as an alternate cation, and nitrate's O atoms could bond with either water's D or the K+.
Compared with tracking nitrogen (N), tracking O would provide a clearer picture of the solution's structure because nitrate's O atoms bond directly with D on water and K+, whereas its N atoms bond only indirectly with them through their bonds with O. In 1982, scientists used neutron diffraction to explore this aqueous solution, but they isotopically labeled nitrate's N atoms. Nobody had ever tried to label nitrate's O atoms because the authors of a renowned book had looked at differences in neutron scattering among O isotopes and concluded they were too small to be useful.
However, Mike Simonson of ORNL knew intense neutron beams could make these differences much more obvious and came up with the idea for the current experiment in the 1990s. It would be more than two decades before advanced instrumentation would be available to make such an experiment possible. At SNS, the world's most intense pulsed, accelerator-based neutron source, researchers recently turned to the NOMAD instrument for neutron diffraction experiments.
"NOMAD allows us to measure a very small difference in scattering between those two solutions," said Joerg Neuefeind, who with ORNL colleague Katharine Page helped make and analyze the measurements. "This difference couldn't be seen without neutrons."
The new measurement revealed that on average 3.9 heavy water molecules bond to each nitrate molecule, a value that is determined with an improved resolution relative to using nitrogen.
Running LAMMPS software on the Oak Ridge Institutional Cluster, Lukas Vlcek tuned a computer simulation to fit the precise, intricate data from the oxygen experiments. Stephan Irle helped interpret the model data, which revealed in high resolution the atomic structure of the solution—that is, how many water molecules surround each oxygen on a nitrate and how many potassium ions also do so. Bonds between the nitrate and water or between the nitrate and potassium constantly swap, and the computational model was able to show that on average two potassium atoms were ionically bound to the nitrate.
Further experimental data is urgently needed to benchmark atomistic simulations, which until now have used data from less precise measurement methods. The inferences drawn from dilute solutions will not be accurate in models that need to predict processes such as scaling, in which minerals clog pipes at industrial refineries. Moreover, learning how to target the ion pairs first spotted in the ORNL study could improve chemical separations for environmental remediation.
Next the researchers will use neutron diffraction to explore solvated molecules significant in mineral formation. That new knowledge may improve fundamental understanding of geochemistry at locations such as the Hanford Site—DOE's largest cleanup effort.
The title of the paper is "Decoding Oxyanion Aqueous Solvation Structure: A Potassium Nitrate Example at Saturation."
More information: Hsiu-Wen Wang et al. Decoding Oxyanion Aqueous Solvation Structure: A Potassium Nitrate Example at Saturation, The Journal of Physical Chemistry B (2018). DOI: 10.1021/acs.jpcb.8b05895
G W Neilson et al. The structure around nitrate ions in concentrated aqueous solutions, Journal of Physics C: Solid State Physics (2002). DOI: 10.1088/0022-3719/15/11/015
Journal information: Journal of Physical Chemistry B
Provided by Oak Ridge National Laboratory