Single-molecule magnetic tweezers reveal dual function of FACT in gene regulation
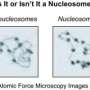
In eukaryotic cells, linear genetic DNA wraps around histones to form stable nucleosomes that further assemble to form chromatins. Nucleosomes represent big barriers to the transcription machine RNA polymerase.
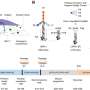
During gene transcription, nucleosomes must first be temporarily removed then rapidly restored afterward. A factor known as Facilitates Chromatin Transcription (FACT) enables the elongation of RNA polymerase on chromatin. But the mechanism by which FACT performs this crucial function is still poorly understood.
Single-molecule magnetic tweezers have become a powerful tool for studying the dynamics of nucleic acid-protein complexes. By exerting tension on a chromatin, magnetic tweezers can be used to study the construction of a chromatin by deconstructing it, thus yielding force spectroscopic fingerprints characteristic of each chromatin. Professors LI Wei and LI Ming from the Institute of Physics of the Chinese Academy of Sciences recently improved the temporal and spatial resolution of their self-developed magnetic tweezers.
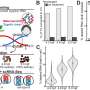
In collaboration with Professors LI Guohong and CHEN Ping from the Institute of Biophysics of the Chinese Academy of Sciences, they investigated the dynamics of nucleosomes and chromatin fibers in the presence of FACT and deciphered the role of FACT in remodeling nucleosomes and chromatin fibers at the single-molecular level. The researchers found that FACT not only destabilized the nucleosome structure to assist the passage of polymerase, but also enhanced the reversibility of nucleosome formation.
In the presence of FACT, nucleosomes are totally unfolded at tensions below 8 pN, which is much lower than for nucleosomes in the absence of FACT (~15 pN). At the same time, nucleosomes cannot reassemble their intact structure in the absence of FACT, due to the dissociation of histones from DNA. However, nucleosomes can reassemble in the presence of FACT, which indicates FACT's additional function of tethering histones to DNA in order to reassemble the intact nucleosome structure.
The dual function of FACT is further proved through coordination between the two subunits SSRP1 and SPT16. These findings provide molecular detail for the interaction between FACT and nucleosomes. These findings suggest that FACT plays an essential role in restringing nucleosomes to preserve the histone on DNA throughout the DNA polymerase passage during DNA transcription.
More information: Ping Chen et al, Functions of FACT in Breaking the Nucleosome and Maintaining Its Integrity at the Single-Nucleosome Level, Molecular Cell (2018). DOI: 10.1016/j.molcel.2018.06.020
Journal information: Molecular Cell
Provided by Chinese Academy of Sciences