Physicists devise method to reveal how light affects materials

Physicists developed a way to determine the electronic properties of thin gold films after they interact with light. Nature Communications published the new method, which adds to the understanding of the fundamental laws that govern the interaction of electrons and light.
"Surprisingly, up to now there have been very limited ways of determining what exactly happens with materials after we shine light on them," says Hayk Harutyunyan, an assistant professor of physics at Emory University and lead author of the research. "Our finding may pave the way for improvements in devices such as optical sensors and photovoltaic cells."
From solar panels to cameras and cell phones—to seeing with our eyes—the interaction of photons of light with atoms and electrons is ubiquitous. "Optical phenomenon is such a fundamental process that we take it for granted, and yet it's not fully understood how light interacts with materials," Harutyunyan says.
One obstacle to understanding the details of these interactions is their complexity. When the energy of a light photon is transferred to an electron in a light-absorbing material, the fphoton is destroyed and the electron is excited from one level to another. But so many photons, atoms and electrons are involved—and the process happens so quickly—that laboratory modeling of the process is computationally challenging.
For the Nature Communications paper, the physicists started with a relatively simple material system—ultra-thin gold layers—and conducted experiments on it.
"We did not use brute computational power," Harutyunyan says. "We started with experimental data and developed an analytical and theoretical model that allowed us to use pen and paper to decode the data."
Harutyunyan and Manoj Manjare, a post-doctoral fellow in his lab, designed and conducted the experiments. Stephen Gray, Gary Wiederrecht and Tal Heilpern—from the Argonne National Laboratory—came up with the mathematical tools needed. The Argonne physicists also worked on the theoretical model, along with Alexander Govorov from Ohio University.
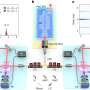
For the experiments, the nanolayers of gold were positioned at particular angles. Light was then shined on the gold in two, sequential pulses. "These laser light pulses were very short in time—thousands of billions of times shorter than a second," Harutyunyan says. "The first pulse was absorbed by the gold. The second pulse of light measured the results of that absorption, showing how the electrons changed from a ground to excited state."
Typically, gold absorbs light at green frequencies, reflecting all the other colors of the spectrum, which makes the metal appear yellow. In the form of nanolayers, however, gold can absorb light at longer wave lengths, in the infrared part of the spectrum.
"At a certain excitation angle, we were able to induce electronic transitions that were not just a different frequency but a different physical process," Harutyunyan says. "We were able to track the evolution of that process over time and demonstrate why and how those transitions happen."
Using the method to better understand the interactions underlying light absorption by a material may lead to ways to tune and manage these interactions.
Photovoltaic solar energy cells, for instance, are currently only capable of absorbing a small percentage of the light that hits them. Optical sensors used in biomedicine and photo catalysts used in chemistry are other examples of devices that could potentially be improved by the new method.
While the Nature Communications paper offers proof of concept, the researchers plan to continue to refine the method's use with gold while also experimenting with a range of other materials.
"Ultimately, we want to demonstrate that this is a broad method that could be applied to many useful materials," Harutyunyan says.
More information: Tal Heilpern et al. Determination of hot carrier energy distributions from inversion of ultrafast pump-probe reflectivity measurements, Nature Communications (2018). DOI: 10.1038/s41467-018-04289-3
Journal information: Nature Communications
Provided by Emory University