Blame the mother's gene: Discovery for a blinding canine eye disease

A gene for canine congenital eye disease has been identified by a collaborative research led by Professor Hannes Lohi's research group in the University of Helsinki. Defective RBP4 leads to vitamin A deficiency and abnormal eye development during pregnancy. The study defines a novel recessive mode of maternal inheritance, which may underlie other types of birth defects.
Collaborating research groups from the University of Helsinki, UC Davis and the University of Jyväskylä describe a new genetic cause for canine congenital eye disease in Irish Soft-Coated Wheaten Terriers. The affected dogs suffer from bilateral microphthalmia i.e. very small eyes and anatomical defects, resulting in incurable blindness. Similar findings are seen in the Microphthalmia-Anophthalmia-Coloboma (MAC) spectrum of human congenital eye malformations, which are important causes of childhood blindness.
"This is the first dog model for MAC disorder and may help to understand the human disease," says DVM Maria Kaukonen, first author of the study and a Ph.D. student in the University of Helsinki.
RBP4 defect leads to vitamin A deficiency—a known risk factor for eye diseases
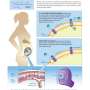
The genetic and functional analyses revealed a deletion mutation in the RBP4 gene. The encoded protein (RBP) binds vitamin A and transfers it from liver to other tissues. Vitamin A transport is essential during the pregnancy, as the lack of this nutrient causes abnormal eye development. During pregnancy, the fetus receives vitamin A through the placenta.
"The mutation changes RBP structure, so that the protein is secreted abnormally from liver and does not function. This greatly reduces RBP and vitamin A levels in bloodstream," says Professor Tom Glaser from UC Davis, a co-corresponding author of the study.
RPB4 has been implicated in human MAC disease. The study demonstrates once again the clinical and genetic similarity of human and canine eye disorders.
Unprecedented mode of maternal inheritance
The study demonstrates a novel recessive mode of genetic inheritance, which has not described before. The researchers discovered that the dam's genotype determines the puppy's disease risk as both the dam and puppy must be homozygous for the mutation to manifest the disease.
"Interestingly, the disease is apparent only when both the puppy and mother are homozygous for the RBP4 mutation. Normally, recessive diseases in mammals depend only on the individual's genetic make-up. Our findings highlight the delicate interaction between the mother and fetus during pregnancy. It is possible that there are similar birth defects with this type of genetic relationship," explains Professor Hannes Lohi, the lead corresponding author of the study.
New genetic test for breeding and diagnostics
As a part of the research, a DNA test has been developed for veterinary diagnosis and breeding purposes. DNA testing is important for breeders to avoid producing more blind dogs. The test can identify carriers and allow better breeding plans. Veterinarians can also use the test for differential diagnosis and to confirm the genetic basis of a suspected condition. So far, the mutation has been found in Irish Soft-Coated Wheaten Terriers but according to Kaukonen it might be present in other breeds too.
More information: Maria Kaukonen et al, Maternal Inheritance of a Recessive RBP4 Defect in Canine Congenital Eye Disease, Cell Reports (2018). DOI: 10.1016/j.celrep.2018.04.118
Journal information: Cell Reports
Provided by University of Helsinki