Connecting hearing helper molecules to the ear bone
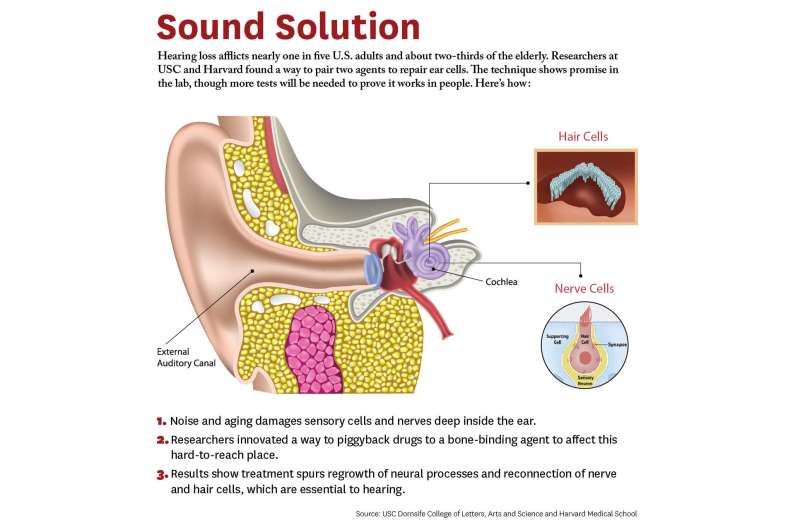
Researchers at USC and Harvard have developed a new approach to repair cells deep inside the ear—a potential remedy that could restore hearing for millions of elderly people and others who suffer hearing loss.
The lab study demonstrates a novel way for a drug to zero in on damaged nerves and cells inside the ear. It's a potential remedy for a problem that afflicts two-thirds of people over 70 years and 17 percent of all adults in the United States.
"What's new here is we figured out how to deliver a drug into the inner ear so it actually stays put and does what it's supposed to do, and that's novel," said Charles E. McKenna, a corresponding author for the study and chemistry professor at USC Dornsife College of Letters, Arts and Sciences. "Inside this part of the ear, there's fluid constantly flowing that would sweep dissolved drugs away, but our new approach addresses that problem. This is a first for hearing loss and the ear. It's also important because it may be adaptable for other drugs that need to be applied within the inner ear."
The paper was published April 4 in the journal Bioconjugate Chemistry. McKenna co-authored it with David Jung of Harvard Medical School, among others. It is the latest achievement in USC's priority program to advance biomedicine, including the recent launching of the USC Michelson Center for Convergent Bioscience. The Michelson Center unites USC experts across disciplines to solve some of the most intractable research challenges related to health at the molecular level. The facility will house the new USC Center of Excellence in Drug Discovery, with McKenna as its director.
There are caveats. The research was conducted on animal tissues in a petri dish. It has not yet been tested in living animals or humans. Yet, the researchers are hopeful given the similarities of cells and mechanisms involved. McKenna says since the technique works in the laboratory, the findings provide "strong preliminary evidence" it could work in living creatures. They are already planning the next phase involving animals and hearing loss.

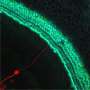
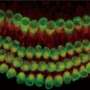
The study breaks new ground because researchers developed a novel drug-delivery method. Specifically, it targets the cochlea, a snail-like structure in the inner ear where sensitive cells convey sound to the brain. Hearing loss occurs due to aging, working with noisy machines and too many loud concerts. Over time, hair-like sensory cells and bundles of neurons that transmit their vibrations break down, as do ribbon-like synapses, which connect the cells.
The researchers designed a molecule combining 7,8-dihydroxyflavone, which mimics a protein critical for development and function of the nervous system, and bisphosphonate, a type of drug that sticks to bones. The pairing of the two delivered the breakthrough solution, the researchers said, as neurons responded to the molecule, regenerating synapses in mouse ear tissue that led to repair of the hair cells and neurons, which are essential to hearing.
"We're not saying it's a cure for hearing loss," McKenna said. "It's a proof of principle for a new approach that's extremely promising. It's an important step that offers a lot of hope."
Hearing loss is projected to increase as the U.S. population ages. Previous research has shown that hearing loss is expected to nearly double in 40 years. Damage to the inner ear can lead to "hidden hearing loss," which is difficulty hearing whispers and soft sounds, especially in noisy places. The new research gives hope to many hoping to avoid loss of hearing and quality of life.
More information: Judith S. Kempfle et al. Bisphosphonate-Linked TrkB Agonist: Cochlea-Targeted Delivery of a Neurotrophic Agent as a Strategy for the Treatment of Hearing Loss, Bioconjugate Chemistry (2018). DOI: 10.1021/acs.bioconjchem.8b00022
Abstract
Hearing loss affects more than two-thirds of the elderly population, and more than 17% of all adults in the U.S. Sensorineural hearing loss related to noise exposure or aging is associated with loss of inner ear sensory hair cells (HCs), cochlear spiral ganglion neurons (SGNs), and ribbon synapses between HCs and SGNs, stimulating intense interest in therapies to regenerate synaptic function. 7,8-Dihydroxyflavone (DHF) is a selective and potent agonist of tropomyosin receptor kinase B (TrkB) and protects the neuron from apoptosis. Despite evidence that TrkB agonists can promote survival of SGNs, local delivery of drugs such as DHF to the inner ear remains a challenge. We previously demonstrated in an animal model that a fluorescently labeled bisphosphonate, 6-FAM-Zol, administered to the round window membrane penetrated the membrane and diffused throughout the cochlea. Given their affinity for bone mineral, including cochlear bone, bisphosphonates offer an intriguing modality for targeted delivery of neurotrophic agents to the SGNs to promote survival, neurite outgrowth, and, potentially, regeneration of synapses between HCs and SGNs. The design and synthesis of a bisphosphonate conjugate of DHF (Ris-DHF) is presented, with a preliminary evaluation of its neurotrophic activity. Ris-DHF increases neurite outgrowth in vitro, maintains this ability after binding to hydroxyapatite, and regenerates synapses in kainic acid-damaged cochlear organ of Corti explants dissected in vitro with attached SGNs. The results suggest that bisphosphonate–TrkB agonist conjugates have promise as a novel approach to targeted delivery of drugs to treat sensorineural hearing loss.
Journal information: Bioconjugate Chemistry
Provided by American Chemical Society