Antibacterial applications of graphene oxides
Bacterial infections are among the greatest threats to human health. However, due to the increasing spread of multidrug-resistant bacteria, the current antibiotic supply appears to be insufficient, thereby necessitating the exploration of novel antibacterial agents. Nano-antibacterial agents represent a new strategy for bacterial treatment. Compared with antibiotics, nano-antibacterial agents have two advantages: (1) broad-spectrum bactericidal effects against Gram-positive and Gram-negative bacteria and (2) long-lasting bactericidal effects due to their extraordinary stability. Significant differences exist in the antibacterial mechanisms between antibiotics and nano-antibacterial agents. Antibiotics can prevent bacterial growth by inhibiting the synthesis of target biomolecules in bacteria, including the cell wall, DNA and proteins. Nano-antibacterial agents kill bacteria through membrane destruction, oxidative stress response, and interactions with cytosolic molecules (lipids, proteins, DNA, etc.).
Graphene oxide (GO) has antibacterial applications. A review titled "Antibacterial Applications of Graphene Oxides: Structure-Activity Relationships, Molecular Initiating Events and Biosafety," published in Science Bulletin, primarily discusses the structure-activity relationships (SARs) involved in GO-induced antibacterial action, the molecular initiating events (MIEs), and the biosafety of antibacterial applications.
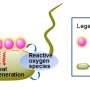
GO possesses a unique two-dimensional (2-D) honeycombed hydrophobic plane structure and hydrophilic groups, including carboxylic (-COOH) and hydroxyl (-OH) groups on its edge, which determine its excellent antibacterial activity. Among these antibacterial mechanisms, this review summarizes the interactions between GO and the bacterial membrane, especially the significant role of MIEs, including redox reactions with biomolecules, mechanical destruction of membranes, and catalysis of extracellular metabolites. The review also discusses in detail the physicochemical effect of GO on the bacterial membrane, such as phospholipid peroxidation, insertion, wrapping and the trapping effect, lipid extraction, and free radicals induced by GO.
Furthermore, this review discusses the effect of size, shape and surface functionality on antibacterial activity to elaborate the SARs, also summarizing the antibacterial nanoproducts that can be used for biomedical, environmental and food engineering applications. The researchers also discuss the biosafety of GO when used in the biomedical field, considering that direct exposure of GO-based antibacterial agents to human cells may induce undesirable hazardous effects. Therefore, scientists must pay close attention to the leakage and release of GO into blood while using GO-coated biomedical devices.
Finally, the review discusses possible future research and the challenges of using GO as a novel nano-antibacterial agent, such as understanding the interactions occurring at GO-bacteria interfaces, the exploration of GO-based nanocomposites to achieve synergistic antibacterial effects, and the immobilization of GO for antibacterial use.
More information: Huizhen Zheng et al, Antibacterial applications of graphene oxides: structure-activity relationships, molecular initiating events and biosafety, Science Bulletin (2017). DOI: 10.1016/j.scib.2017.12.012
Provided by Science China Press