A nonaddictive opioid painkiller with no side effects

What if scientists could develop an opioid-based painkiller that is not addictive and has limited side effects?
That is possible based on new findings by an international team of scientists that included contributions from top researchers at the USC Michelson Center for Convergent Bioscience.
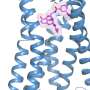
The international team captured the crystal structure of the kappa opioid receptor—critical for providing pain relief—in action on the surface of human brain cells. The researchers also made another important discovery: a new opioid-based compound that, unlike current opioids, activates only the kappa opioid receptor, raising hopes that they may develop a painkiller that has no risk of addiction and, therefore, none of the devastating consequences and side effects that accompany it.
The findings were published Jan. 4 in the journal Cell. They are an example of how USC Michelson Center scientists collaborate with a range of experts in multiple disciplines to conduct groundbreaking research, including the opioid addiction.
First, do no harm
The current challenge facing scientists in drug research and development is twofold: develop new alternatives to ease pain while minimizing side effects. Amid an opioid addiction crisis, this is a tall order, and it is urgent. More than 1 in 10 Americans suffer chronic pain, according to the National Institutes of Health. At the same time, millions of Americans are addicted to opioids.
Captained by researchers at the University of North Carolina at Chapel Hill, the team of 24 scientists on this latest study included three USC Michelson Center scientists who are among the most-recognized names in research on the special receptors found on the surface of the neuron: Raymond C. Stevens, Vadim Cherezov and Vsevolod "Seva" Katritch. All three are affiliated with the USC Dornsife College of Letters, Arts and Sciences.
The G protein-coupled receptors, found on the surface of the membrane, are the gatekeepers of communication with cells and are therefore the intended target of most therapeutics. The solution to pain, disease and other conditions begins with understanding—and seeing very clearly—the structure of the receptors when they are inactive and when they are active, interacting with a drug compound.
Typically, scientists determine the structure of receptors by forcing the proteins into a crystal lattice that they then expose to X-rays. Essentially, they want to create an accurate model of the receptor when it does and does not interact with a drug compound.
However, these G protein-coupled receptors are challenging to capture in a stabilized state with traditional X-ray crystallography. Like ill-behaving toddlers, they are highly dynamic, move frequently and very fragile. That is why Stevens, Cherezov and Katritch developed some breakthrough techniques for this special class of proteins that have led to more accurate crystallography.
At a cellular level, their work has led to a greater understanding of the receptors and their behaviors. From a holistic perspective, their research is explaining how humans respond to drugs. Furthermore, they have set the foundation for a new wave of therapeutics that are much more precisely targeted than their predecessors to address illnesses and conditions with fewer unintended side effects.
The power of three
Stevens is a molecular biologist and chemist hailed as a pioneer in solving the structures of G protein-coupled receptors. He developed a method of experimenting known as "high-throughput crystallography" in structural biology, which uses robotics, data processing and management software, as well as liquid handling devices and detectors to conduct millions of tests.
Cherezov is a structural biologist who developed new ways to herd capricious membrane proteins like the G protein-coupled receptors into well-behaved crystals. He uses lipids similar to those found in cell membranes to form a special "cubic phase." This technique ensures that the receptors behave as if they had never left their home on the membrane, even as they form crystals.
The Lipidic Cubic Phase technology has been successfully applied to a majority of the GPCRs that have been solved, including the previous kappa opioid receptor structure in its inactive state, according to Cherezov. By adding a stabilizing nanobody, the researchers are able to capture the structure in a fully active state, he said.
Katritch, a biophysicist and computational biologist, has developed computer models of receptor interactions with ligands that activate or inactivate receptors. This allows scientists to quickly test the receptor interactions with millions of ligands in a virtual lab—his computer—so that they may select the molecules that have the most beneficial therapeutic properties for more testing.
In the case of the kappa opioid receptor, his computer analyses enabled the scientists to modify the chemistry of the ligands so that eventually they developed ligands that affected only the kappa opioid receptor.
Currently, most opioids bind to several opioid receptors on the membrane of brain cells, which has its share of drawbacks. They alleviate pain but cause a range of side effects, from nausea to numbness, constipation, anxiety, severe dependency, hallucinations and even death by respiratory depression.
In this study, the computer models revealed the formulations that would create the strongest bond between the ligand and the kappa opioid receptor without affecting other receptors.
Katritch said the latest research may pave the way for a major drug breakthrough.
"We have already found the structure of the inactive kappa opioid receptor highly useful for discovering potential candidates for a new painkiller," Katritch said. "Now with the structure of the active receptor, we have a template for designing new types of pain medications that have no disruptive side effects for patients and would reduce the burden that opioid addiction has placed on society."
More information: Tao Che et al. Structure of the Nanobody-Stabilized Active State of the Kappa Opioid Receptor, Cell (2018). DOI: 10.1016/j.cell.2017.12.011
Journal information: Cell
Provided by University of Southern California