Researchers explore how atoms align in magnetic materials
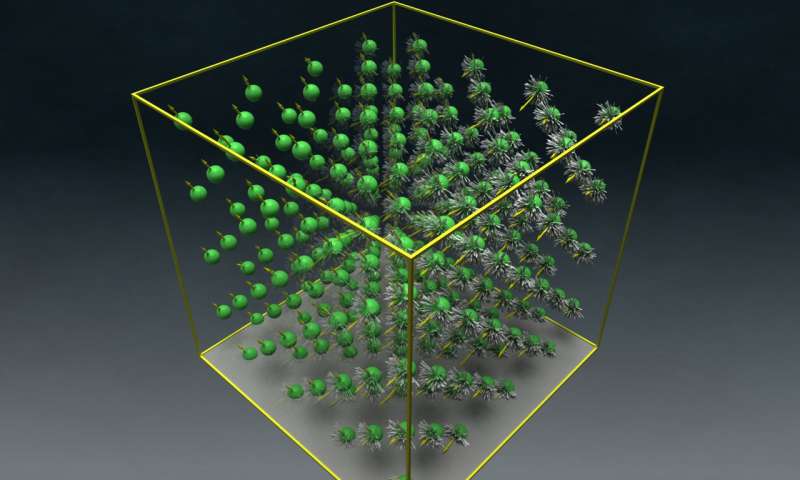
The atoms inside materials are not always perfectly ordered, as usually depicted in models. In magnetic, ferroelectric (or showing electric polarity) and alloy materials, there is competition between random arrangement of the atoms and their desire to align in a perfect pattern. The change between these two states, called a phase transition, happens at a specific temperature.
Markus Eisenbach, a computational scientist at the Department of Energy's Oak Ridge National Laboratory, heads a group of researchers who've set out to model the behavior of these materials using first principles – from fundamental physics without preset conditions that fit external data.
"We're just scratching the surface of comprehending the underlying physics of these three classes of materials, but we have an excellent start," Eisenbach says. "The three are actually overlapping in that their modes of operation involve disorder, thermal excitations and resulting phase transitions – from disorder to order – to express their behavior."
Eisenbach says he's fascinated by "how magnetism appears and then disappears at varying temperatures. Controlling magnetism from one direction to another has implications for magnetic recording, for instance, and all sorts of electric machines – for example, motors in automobiles or generators in wind turbines."
The researchers' models also could help find strong, versatile magnets that don't use rare earth elements as an ingredient. Located at the bottom of the periodic table, these 17 materials come almost exclusively from China and, because of their limited source, are considered critical. They are a mainstay in the composition of many strong magnets.
Eisenbach and his collaborators, which includes his ORNL team and Yang Wang with the Pittsburgh Supercomputing Center, are in the second year of a DOE INCITE (Innovative and Novel Computational Impact on Theory and Experiment) award to model all three materials at the atomic level. They've been awarded 100 million processor hours on ORNL's Titan supercomputer and already have impressive results in magnetics and alloys. Titan is housed at the Oak Ridge Leadership Computing Facility (OLCF), a DOE Office of Science user facility.
The researchers tease out atomic-scale behavior using, at times, a hybrid code that combines Wang-Landau (WL) Monte Carlo and locally self-consistent multiple scattering (LSMS) methods. WL is a statistical approach that samples the atomic energy landscape in terms of finite temperature effects; LSMS determines energy value. With LSMS alone, they've calculated the ground state magnetic properties of an iron-platinum particle. And without making any assumption beyond the chemical composition, they've determined the temperature at which copper-zinc alloy goes from a disordered state to an ordered one.
Moreover, Eisenbach has co-authored two materials science papers in the past year, one in Leadership Computing, the other a letter in Nature, in which he and colleagues reported using the three-dimensional coordinates of a real iron-platinum nanoparticle with 6,560 iron and 16,627 platinum atoms to find its magnetic properties.
"We're combining the efficiency of WL sampling, the speed of the LSMS and the computing power of Titan to provide a solid first-principles thermodynamics description of magnetism," Eisenbach says. "The combination also is giving us a realistic treatment of alloys and functional materials."
Alloys are comprised of at least two metals. Brass, for instance, is an alloy of copper and zinc. Magnets, of course, are used in everything from credit cards to MRI machines and in electric motors. Ferroelectric materials, such as barium titanate and zirconium titanate, form what's known as an electric moment, in a transition phase, when temperatures drop beneath the ferroelectric Curie temperature – the point where atoms align, triggering spontaneous magnetism. The term – named after the French physicist Pierre Curie, who in the late 19th century described how magnetic materials respond to temperature changes – applies to both ferroelectric and ferromagnetic transitions. Eisenbach and his collaborators are interested in both phenomena.
Eisenbach is particularly intrigued by high-entropy alloys, a relatively new sub-class discovered a decade ago that may hold useful mechanical properties. Conventional alloys have a dominant element – for instance, iron in stainless steel. High-entropy alloys, on the other hand, evenly spread out their elements on a crystal lattice. They don't get brittle when chilled, remaining pliable at extremely low temperatures.
To understand the configuration of high-entropy alloys, Eisenbach uses the analogy of a chess board sprinkled with black and white beads. In an ordered material, black beads occupy black squares and white beads, white squares. In high-entropy alloys, however, the beads are scattered randomly across the lattice regardless of color until the material reaches a low temperature, much lower than normal alloys, when it almost grudgingly orders itself.
Eisenbach and his colleagues have modelled a material as large as 100,000 atoms using the Wang-Landau/LSMS method. "If I want to represent disorder, I want a simulation that calculates for hundreds if not thousands of atoms, rather than just two or three," he says.
To model an alloy, the researchers first deploy the Schrodinger equation to determine the state of electrons in the atoms. "Solving the equation lets you understand the electrons and their interactions, which is the glue that holds the material together and determines their physical properties."
All of a material's properties and energies are calculated by many hundreds of thousands of calculations over many possible configurations and over varying temperatures to give a rendering so that modelers can determine at what temperature a material loses or gains its magnetism, or at what temperature an alloy goes from a disordered state to a perfectly ordered one.
Eisenbach eagerly awaits the arrival of the Summit supercomputer – five to six times more powerful than Titan – to OLCF in late 2018. "Ultimately, we can do larger simulations and possibly look at even more complex disordered materials with more components and widely varying compositions, where the chemical disorder might lead to qualitatively new physical behaviors."
Journal information: Nature
Provided by US Department of Energy





















