Chemists suggest a new way to synthesize steroid analogs
Scientists from RUDN University and the Ivan Franko National University of Lviv have found a way to produce aromatic rings in organic compounds in three stages. These stages proceed successively in one-pot conditions and at room temperature. Now analogues of hormones, steroids, some sugars, terpenes and other complex organic substances can be synthesized faster and at softer conditions. The paper was published in Tetrahedron Letters.
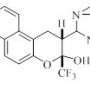
Steroids are substances with high biological activity. They include anti-inflammatory drugs (e.g., dexamethasone), as well as some hormones. Moreover, steroids are used to produce vitamin D. Seven Nobel Prizes were awarded between 1927 and 1975 for studying these important substances. Synthesizing steroids is not always easy, because they are based on a polysubstituted tetracyclic system. Chemists from RUDN University and their Ukrainian colleagues found a new way to produce steroids and other substances that contain six-membered rings.
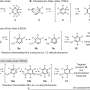
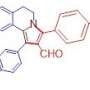
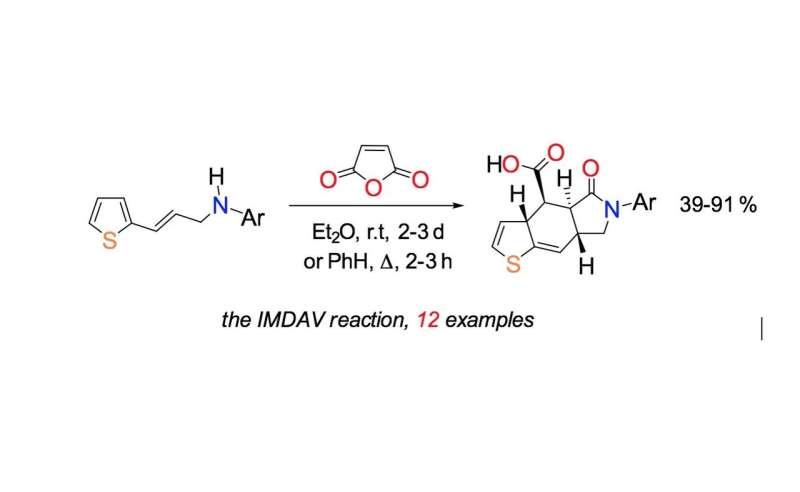
"The signature moment of the synthetic part is a tandem cascade of reactions—the reactions proceed successively in the same flask under the same reaction conditions: acylation of the nitrogen atom of the starting 3-thienylallylamine with maleic anhydride, the intramolecular Diels-Alder reaction and subsequent aromatization of the cycloaddition adduct," says Fedor Zubkov, assistant professor of the Department of Organic Chemistry at the RUDN University.
Diene synthesis (or the Diels-Alder reaction) is one of the most popular reactions in organic chemistry. It involves synchronous formation of two sigma-double bonds from one double bond of dienophile and two double bonds of diene; the main product is a six-membered ring with one double bond. Otto Diels and Kurt Alder received the Nobel Prize in 1950 for this reaction. Another Nobel laureate, Robert Woodward, was inspired and bound his life with chemistry after reading about Diels-Alder's reaction. Later, he returned to it in his studies several times. Curiously enough, it was Robert Woodward who founded the Tetrahedron Letters journal, which published this paper by Russian scientists.
The intramolecular Diels-Alder reaction runs inside one molecule. With this reaction, complex molecules including six-membered rings can be synthesized in one step. Chemists can control the direction of how the reaction goes and the bonds involved in it. By regulating the conditions, they can obtain exactly the necessary isomers—the substances with the same number of atoms in the molecule, but with different structural formulas. The scientists found out that vinylthiophenes are suitable for this task.
"We can see that six-membered cycles are formed—the structural motifs that are most often found in natural biologically active compounds (alkaloids, sugars, hormones, steroids, terpenes and others). As a rule, the interaction between diene and dienophile (the constituents of diene synthesis) does not require severe temperature conditions or the use of catalysts, and the products formed are very pure and easily isolated," Fedor Zubkov comments.
Earlier, according to Fedor Zubkov, synthesis of the similar substances required a temperature of 160 °C to produce the desired six-membered ring. In the new study, this process takes two to three days at room temperature. The reaction runs more quickly in boiling benzene (at 80 ° C)—it takes only two hours. In this case, however, the final product is contaminated with impurities. The process runs in three stages and the yield of desired reaction products is 39 to 91 percent, which is enough to synthesize hormone-like drug components and many other chemicals more quickly, more conveniently and more efficiently than before.
More information: Yuriy I. Horak et al, The intramolecular Diels-Alder vinylthiophen (IMDAV) reaction: An easy approach to thieno[2,3- f ]isoindole-4-carboxylic acids, Tetrahedron Letters (2017). DOI: 10.1016/j.tetlet.2017.09.038
Provided by RUDN University