Scientists propose method to increase the efficiency of solar batteries
Researchers from Department of Material Sciences, Lomonosov MSU, have determined how changing the ratio of components forming the light-absorbing layer of a perovskite solar cell influences the structure of resulting films and battery efficiency. The results of the study were published in the Journal of Physical Chemistry C.
Organic-inorganic perovskites are a new class of photoactive (i.e. reacting to light) materials. They were named after the mineral perovskite (CaTiO3, calcium titanate) because of structural similarities, though their own is much more interesting. Such materials can be used to create perovskite solar batteries, which were first introduced only five years ago, but have already surpassed the efficiency of more expensive silicon solar elements.
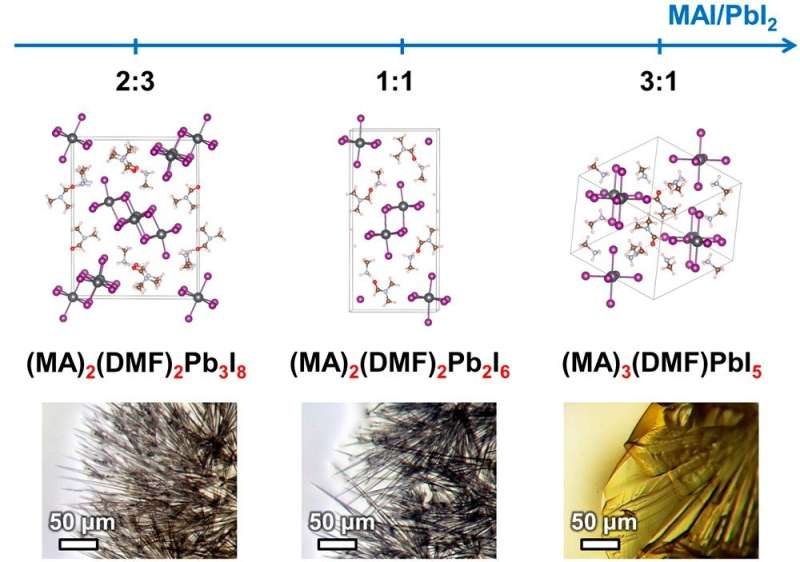
In their previous study, the authors have found that filiform (wire-like) hybrids of perovskites have acquired their shape because of the structure of intermediate compounds, which are formed during the process of perovskite crystallization. The researchers have discovered a whole group of these compounds, every one of which is a crystalline solvate. The crystalline solvates are crystalline compounds with molecules of the precursor components' solvent built into their structure. The dissolved components precipitate from the solution and form a crystalline film of perovskite.
The researchers selected and described three intermediate compounds which are crystalline solvates of one of the two solvents used most often in creating perovskite solar batteries. For two of these compounds, their crystal structure was established for the first time.
"We have found that the the formation of intermediate compounds is one of the key factors that determines the functional properties of the final perovskite layer because perovskite crystals inherit the shape of those compounds. This, in turn, influences the film morphology and solar cell efficiency. It is especially important when creating thin perovskite films, because the needle-like or filiform shape of crystals will lead to the film being discontinuous, which will significantly lower the efficiency of the solar cell. The knowledge on the influence of the ratio of precursor reagents on the shape of the final perovskite crystals will allow researchers to deliberately choose the conditions for obtaining optimal films, which will result in perovskite cells with high efficiency," says principal investigator Alexey Tarasov of Lomonosov Moscow State University.
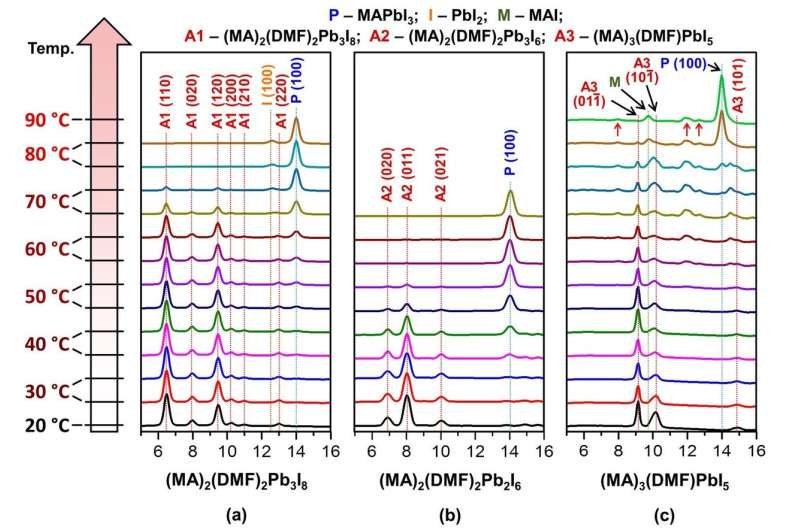
Such intermediate compounds are unstable, so the authors used synchrotron radiation and low temperatures to cool the crystals to the temperature of -173 °C. The freezing allowed the scientists to stop the decomposition of the crystals and to run the necessary measurements to determine the structure of the solvates.
Additionally, the researchers have studied the thermal stability of the obtained compounds and managed to calculate the energy of their formation using quantum-chemical modeling. Knowing the formation energy allows to explain why certain crystals are formed when using different solvents.
The authors have also learned that the ratio of reagents in solution specifically determines which intermediate compound will form in the process of crystallization. Crystal structure of the intermediate compound defines the shape of the perovskite crystals formed, which determines the structure of the light-absorbing layer. This structure, in turn, influences the output of the solar battery created.
More information: Andrey A. Petrov et al, Crystal Structure of DMF-Intermediate Phases Uncovers the Link Between CH3NH3PbI3 Morphology and Precursor Stoichiometry, The Journal of Physical Chemistry C (2017). DOI: 10.1021/acs.jpcc.7b08468
Journal information: Journal of Physical Chemistry C
Provided by Lomonosov Moscow State University