Researchers test the capability of a novel nanoparticle to remove cadmium toxicity from a freshwater system
Nanotechnology plays an important role in removing toxic chemicals found in the soil. Currently more than 70 Environmental Protection Agency (EPA) Superfund sites are using or testing nanoparticles to remove or degrade environmental contaminants. One of these—nano-zero-valent iron—is widely used, though its effect on organisms has not been examined.
In a recent experiment, a team of scientists from UC Santa Barbara tested the effect of sulfurized nano-zero-valent iron (FeSSi) on a common freshwater alga (Chlamydomonas reinhardtii). They found that FeSSi picked up cadmium from a watery medium and alleviated cadmium toxicity to that alga for more than a month. Their results appear in the journal ACS Nano.
"However, when FeSSi was doing what it was designed to do, we found it was up to 10 times more toxic when bound to cadmium than without," said lead author Louise Stevenson, a postdoctoral scholar in UCSB's Department of Ecology, Evolution and Marine Biology (EEMB). "The current standards for what is an acceptable concentration to use are based on data from the particle itself unbound to the contaminant. Our work suggests that those allowable limits potentially could be huge underestimations of the actual toxicity."
To simulate a precipitation event in which toxic material from soil washes into a waterway, the researchers dosed C. reinhardtii with the cadmium-laced FeSSi and waited an hour before taking measurements. They found that organic material the algae itself produced as a byproduct of photosynthesis mitigated the toxicity of FeSSi and allowed the nanoparticle to remediate up to four times as much cadmium.
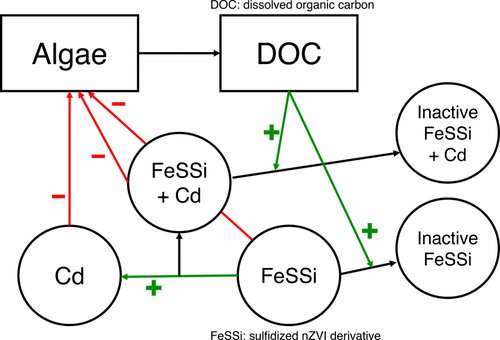
"The organic material makes the FeSSi particle less toxic, which allows a greater zone of remediation and increases the cadmium concentrations that can be used," Stevenson said. "That's interesting because every natural system contains some organic material. Along with the toxic effect of the nanoparticles just on cell viability, we identified an important feedback between organic materials produced by the algae itself decreasing toxicity, which decreases toxicity to the algae.
According to Stevenson, the environmental effects of nanotechnology are very context specific, making overarching predictions difficult. So, the UCSB team designed a dynamic ecological model that can be used to extrapolate what they empirically tested. The investigators amassed enough data to develop a series of equations to describe the dynamics of the concentrations they tested.
"We're developing new technology faster than we can predict its environmental impact," Stevenson noted. "That makes it very important to design experiments that are ecologically and environmentally relevant but also get at dynamics that can be extrapolated to other systems."
More information: Louise M. Stevenson et al. Remediation of Cadmium Toxicity by Sulfidized Nano-Iron: The Importance of Organic Material, ACS Nano (2017). DOI: 10.1021/acsnano.7b05970
Journal information: ACS Nano
Provided by University of California - Santa Barbara
















