Bacterial cell wall changes produce more fatty molecules
Bacteria and other microbes can pump out building blocks for biofuels. Unfortunately, getting microbes to produce enough building blocks, lipids, presents a significant challenge. Microbes often fall short of what they could theoretically produce. This study examined high-lipid mutant variants of Rhodobacter sphaeroides. The scientists showed that altering the bacterial cell envelope results in more lipids relative to the parent strain.
Knowledge of the mechanisms that limit microbial lipid production can reveal new strategies to increase lipid yield. The result? Increased economic viability of alternatives to fuels or chemicals currently derived from petroleum.
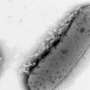
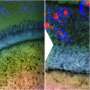
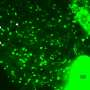
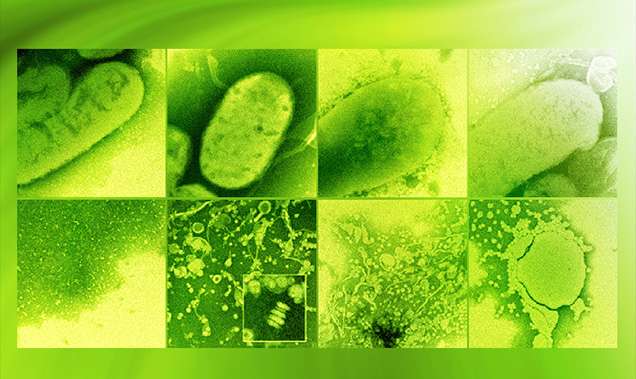
Microbial lipids are potential replacements for petroleum-based fuels and chemicals; however, their production often falls short of theoretical yield, and strategies for improvement are needed. Researchers from the U.S. Department of Energy (DOE) Great Lakes Bioenergy Research Center; the University of Wisconsin-Madison; and the Environmental Molecular Sciences Laboratory (EMSL), a DOE Office of Science user facility, built upon their research on microbial lipid production by examining a new class of Rhodobacter sphaeroides mutants that exhibited enhanced lipid accumulation relative to the parent strain. The researchers used the FEI Tecnai T-12 cryo-transmission electron microscope and structured illumination super-resolution fluorescence microscope at EMSL.
They determined the chemical sensitivity profiles indicated high-lipid mutants were sensitive to drugs that target the cell envelope. They also observed changes in cell shape, suggesting previously undescribed alterations in the bacterial cell envelope could be used to increase lipid production. Importantly, a subset of the high-lipid mutants secreted lipids. Two mutants accumulated approximately 60 percent of their total lipids extracellularly, potentially enabling easy recovery of a product from a bioreactor.
When one of the highest lipid-secreting strains was grown in a fed-batch bioreactor, its lipid content was comparable to oleaginous microbes, defined as those accumulating 20 percent or more of their dry cell weight as lipid. Knowledge of the biological mechanisms that limit lipid production can inform new genetic engineering and growth strategies and allow this important class of molecules to be adopted as fuels or chemicals on a larger scale.
More information: Kimberly C. Lemmer et al. Mutations That Alter the Bacterial Cell Envelope Increase Lipid Production, mBio (2017). DOI: 10.1128/mbio.00513-17
Journal information: mBio
Provided by US Department of Energy