August 1, 2017 report
Visual processing capabilities of flatworm found to be more complex than thought

(Phys.org)—A team of researchers with the National Centre for Biological Sciences in India has found that the visual processing capabilities of the planarian flatworm are much more complex than has been thought. In their paper published on the open access site Science Advances, the team describes a series of experiments they carried out with the flatworm and what they learned about its visual processing capabilities.
Planarian flatworms are common around the globe. They are shaped somewhat like an eel with a flattened body and head. Most live in water, fresh or salty and some have a pair of dark eyes on their head. Planarian flatworms are also very well known in the biological world due to their unique ability to regenerate body parts—if you split one in two, for example, both parts will grow into a complete flatworm. Perhaps even more impressive, the flatworm can even regrow its head if lost. Prior research with the flatworms has shown that they are averse to light. In this new effort, the researchers sought to learn more about how flatworm vision works because of their ability to regenerate eyes when the head is lost.
The team started by repeating prior experiments involving shining both white and UV light on test flatworms—they found that as expected, the flatworms swam away from either type. But they also found the flatworms disliked some colors less than others. When they had to choose between exposure to green or blue light, the flatworms chose green; they also chose red over green. This finding was surprising, considering the eyes of the flatworm are little more than cups containing photoreceptors—and their brains are little more than a few neurons. A closer look showed the flatworms were not actually able to make out the difference in color, but were responding instead to the brightness of the hues.

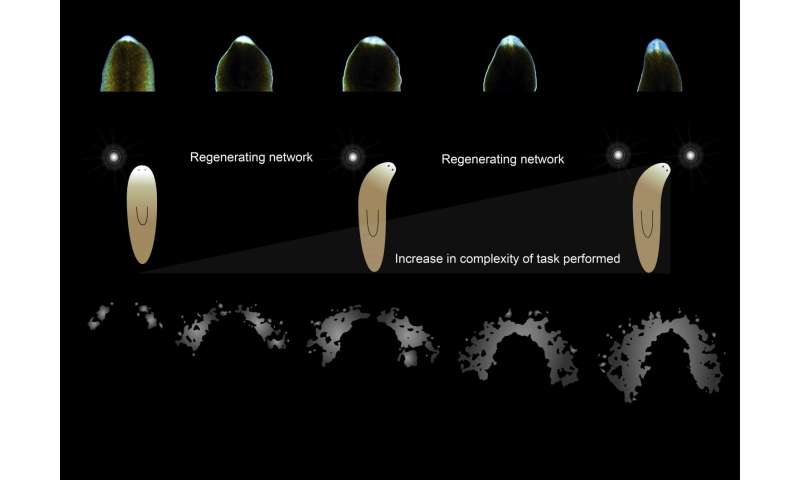
The researchers also cut off the heads of specimens and tested them during the regeneration period—they were surprised to find the headless flatworms still shied away from UV light, suggesting light sensitivity in the skin. They also found that although the eyes regenerated after just four days, the flatworms were not able to respond to light again until a full week had passed; color preference did not appear again until after 12 days.
-
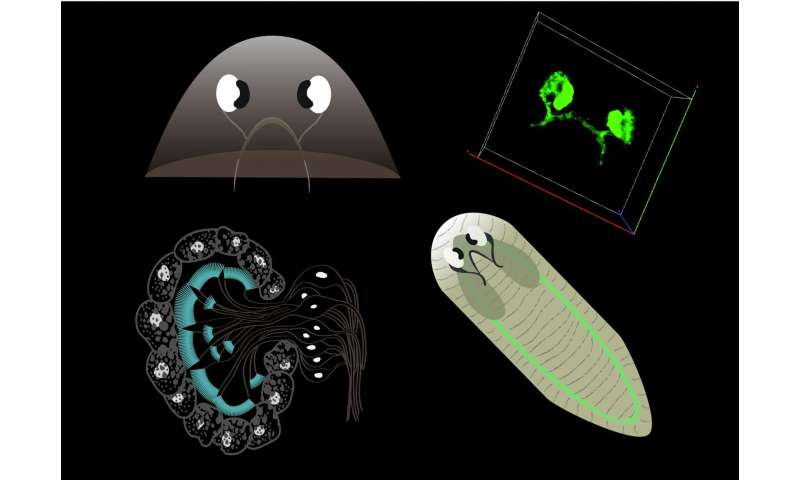
Schematic image of the planarian visual network. Planarians form the optic chiasm and visual axons project into the visual center for transmitting light information to the brain. The components of the planarian photoreceptor. The multicellular pigment cup produced by apposition of several pigment cells. Optic chiasm stained with a photoreceptor neuron marker. Scheme of planarian nervous system. Credit: Akash Gulyani Lab, inStem, Bangalore, India -
Schematics showing time line of return of different phototactic abilities during head regeneration in planarians. Few days after regeneration, worms sense light but have no ability to discriminate wavelengths, which is acquired gradually significantly later. Credit: Akash Gulyani Lab, inStem, Bangalore, India
More information: Nishan Shettigar et al. Hierarchies in light sensing and dynamic interactions between ocular and extraocular sensory networks in a flatworm, Science Advances (2017). DOI: 10.1126/sciadv.1603025
Abstract
Light sensing has independently evolved multiple times under diverse selective pressures but has been examined only in a handful among the millions of light-responsive organisms. Unsurprisingly, mechanistic insights into how differential light processing can cause distinct behavioral outputs are limited. We show how an organism can achieve complex light processing with a simple "eye" while also having independent but mutually interacting light sensing networks. Although planarian flatworms lack wavelength-specific eye photoreceptors, a 25 nm change in light wavelength is sufficient to completely switch their phototactic behavior. Quantitative photoassays, eye-brain confocal imaging, and RNA interference/knockdown studies reveal that flatworms are able to compare small differences in the amounts of light absorbed at the eyes through a single eye opsin and convert them into binary behavioral outputs. Because planarians can fully regenerate, eye-brain injury-regeneration studies showed that this acute light intensity sensing and processing are layered on simple light detection. Unlike intact worms, partially regenerated animals with eyes can sense light but cannot sense finer gradients. Planarians also show a "reflex-like," eye-independent (extraocular/whole-body) response to low ultraviolet A light, apart from the "processive" eye-brain–mediated (ocular) response. Competition experiments between ocular and extraocular sensory systems reveal dynamic interchanging hierarchies. In intact worms, cerebral ocular response can override the reflex-like extraocular response. However, injury-regeneration again offers a time window wherein both responses coexist, but the dominance of the ocular response is reversed. Overall, we demonstrate acute light intensity–based behavioral switching and two evolutionarily distinct but interacting light sensing networks in a regenerating organism.
Journal information: Science Advances
© 2017 Phys.org