A new molecular scissors act like a GPS to improve genome editing
Researchers from the University of Copenhagen (Denmark), led by the Spanish researcher Guillermo Montoya, have discovered how Cpf1, a new molecular scissors, unzips and cleaves DNA. This member of the CRISPR-Cas family displays high accuracy, and is capable of acting like a GPS in order to identify its destination within the intricate map of the genome. The high precision of Cpf1 will improve the use of this type of technology in repairing genetic damage and in other medical and biotechnological applications.
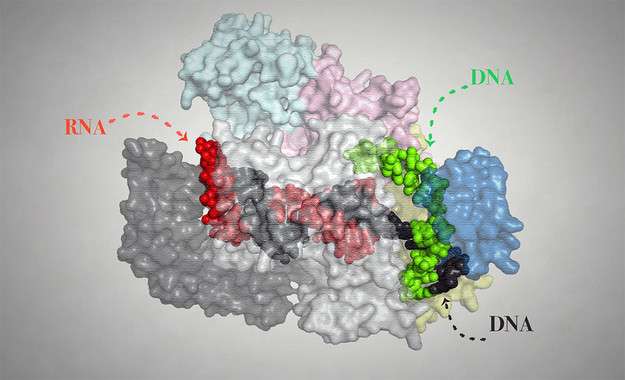
The team succeeded in visualizing and describing how Cpf1 works. This protein belongs to the Cas family and enables the cleavage of double-stranded DNA, thus allowing the initiation of the genome modification process. The results of the study have been published in the journal Nature.
Guillermo Montoya, a researcher in the fields of biochemistry and molecular biology who led the study, explains to SINC that the new molecular scissors "will enable us to more safely modify and edit the instructions written in the genome, due to the utmost precision of the target DNA sequence recognition."
The CRISPR Cas9 system for cutting and pasting genome sequences is already being used to modify animal and plant genomes. It is also used to treat illnesses such as cancer and retinal diseases in humans, and its applications are increasing.
X-Ray Crystallography Technique
Researchers around the world are trying to perfect this genome editing technique with the aim of making it yet more precise and efficient. To achieve this, they have also focused on other proteins that specifically cut DNA, such as Cpf1, whose manipulation can direct them to specific locations in the genome. Montoya's team has achieved this using an X-ray crystallography to decipher the molecular mechanisms controlling this process.

"We radiated the crystals of the Cpf1 protein using X-rays to be able to observe its structure at atomic resolution, enabling us to see all its components," says the co-author of this study. "X-ray diffraction is one of the main biophysical techniques used to elucidate biomolecular structures.
"The main advantage of Cpf1 lies in its high specificity and the cleaving mode of the DNA, since it is possible to create staggered ends with the new molecular scissors, instead of blunt-ended breaks as is the case with Cas9, which facilitates the insertion of a DNA sequence.
"The high precision of this protein recognising the DNA sequence on which it is going to act functions like a GPS, directing the Cpf1 system within the intricate map of the genome to identify its destination. In comparison with other proteins used for this purpose, it is also very versatile and easily reprogrammed," Montoya adds.
Genetic diseases and tumours
These properties make this system "particularly suitable for its use in the treatment of genetic diseases and tumours," he says.
The team has previously worked with the French biotechnology company Celletics on the use of meganucleases—other proteins that can be redesigned to cut the genome in a specific location—to treat certain types of leukemia.
The new technology "can also be used to modify microorganisms, with the aim of synthesising the metabolites required in the production of drugs and biofuels," adds Montoya.
More information: Stefano Stella et al. Structure of the Cpf1 endonuclease R-loop complex after target DNA cleavage, Nature (2017). DOI: 10.1038/nature22398
Journal information: Nature
Provided by Plataforma SINC