Wastewater nutrients converted into clean fertiliser

Aalto University's water and environmental technology laboratory gives off an undeniable whiff of the sewer – and this stench has its roots in a failed West African recycling business.
While working in Ghana three years ago, postdoctoral researcher Surendra Pradhan had to acknowledge that his wastewater business made little sense.
"Recycling is always commendable, but rarely economically viable. We launched several projects only to discover that it was surprisingly difficult to sell our product. The average farmer did not utilise compost and there were few buyers for slurry containing human waste in particular. I finally came to the conclusion that the most sensible option was to manufacture something for which there already was a market."
Escaping a senseless cycle
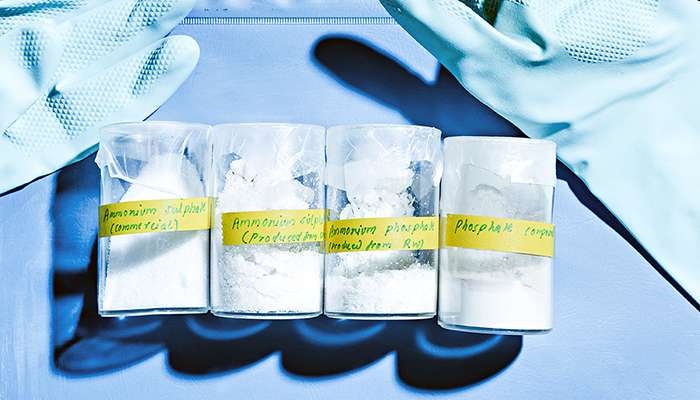
Pradhan started researching the matter and discovered that the best way to utilise wastewater nitrogen was to recover it in ammonium sulphate form, which is used in fertilisers and does not contain residual chemicals and pharmaceuticals that are often present in slurry. As a bonus, the same process can recover another agent of eutrophication, phosphorus, which up to now has, like nitrogen, mostly been a problem that wastewater treatment facilities must remove.
Traditionally, the fertiliser nitrogen needed for ammonium sulphate has been made using the Haber-Bosch method, a process developed by German scientists one hundred years ago that converts atmospheric nitrogen into ammonia. The process requires high temperatures and pressures, and is thus quite a glutton for energy: the manufacture of fertiliser nitrogen accounts for an estimated two percent of global energy consumption.
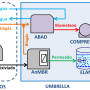
Pradhan and his researcher colleagues at Aalto University developed a nutrient capturing method that conducts wastewater into a reactor where its pH value is raised with calcium hydroxide so that the water's ammoniacal nitrogen is converted into gaseous ammonia. For its part, the ammonia is separated via a semi-permeable membrane after which it is dissolved into sulphuric acid to create ammonium sulphate. The phosphorus in the water is precipitated with calcium salts and can then be utilised as fertiliser.
"As calcium is, alongside nitrogen and phosphorus, the most important nutrient required for growing crops in the acid soil of Finland, using calcium-derived substances to elevate pH levels does not increase costs because it just winds up being a part of the end product," says postdoctoral researcher Anna Mikola.
Testing of the method began with a smallish container version and has now progressed to a slightly larger reactor. The results are promising. Experiments have managed to separate up to 99% of the nitrogen and 90-99% of the phosphorus in urine, even though the method consumes considerably less energy and chemicals than the commonly employed nitrification-denitrification wastewater treatment process.
"In it the nitrogen, whose recovery into fertiliser form consumes an enormous amount of energy, is lost to the winds. The process also accounts for the lion's share of the energy required for water purification. Our aim is to escape this senseless cycle," Mikola says.
From small container to trailer and a pilot
A patent application for the method is currently being processed, but there's still a ways to go before it is adopted into industrial use. Professor Riku Vahala says the team prefers to take baby steps forward rather than barging into a wall at full speed.
"Too often great expectations lead to a direct leap from the laboratory to the factory, which eats up all the money and erodes faith in the entire technology. We're proceeding first from a small container scale to a trailer-sized application and, if all goes well, we'll be prepared to build a demonstration facility in two years time."
Before this, the researchers need to find out how, for example, the method works with various types of wastewater – the kinds produced by households, biogas plants and, say, marinas are radically different, after all. The next experiments are to be carried out on reject water created in conjunction with the drying of sludge at the Helsinki Region Environmental Services Authority's Viikinmäki wastewater treatment facility's sludge digestion chamber as well as at Gasum biogas plants.
The huge significance of water treatment adds to the challenge.
"When dealing with an issue that has an impact on the environment and human health, the involved parties do not want to take any chances and instead prefer to employ old familiar methods, even if they are more expensive and less effective," Vahala sums up.
The way in which the method will be integrated with operations at facilities also remains open. The trend in industry is to focus tightly on the core business and, accordingly, wastewater treatment has often been outsourced. In addition, many municipal wastewater treatment facilities have transferred responsibility for the further processing of sludge to external companies. Pradhan, Mikola and Vahala believe that, once the technology is finished, the best way to introduce it as broadly as possible is via third parties.
"It makes no sense for each large or small waste treatment facility to also become a fertiliser seller. It would be much wiser for one or more firms to commercialise our technology, operations and also the end product it yields," Mikola reckons.
Good for the Baltic Sea
The method has other positive environmental impacts in addition to energy savings. Researchers estimate that recovering nutrients from community wastewater enables the replacement of six percent of industrially produced ammoniacal nitrogen and ten percent of the phosphorus used as fertiliser. At present, fertiliser phosphorus is excavated from mines, which threaten to run dry over the next century unless recovery becomes more efficient. At treatment facilities, however, phosphorus ends up in sludge, which is mainly used as a growth medium for grass and road shoulders because food production regulations and image concerns hamper its utilisation as a crop nutrient.
Effective and economically-attractive recovery would also benefit the Baltic Sea and other water systems in Finland. The removal of phosphorus from community wastewater is in good shape in Finland, but when it comes to nitrogen, the differences between treatment facilities and localities are great. For the most part, Finnish legislation places an obligation to conduct enhanced nitrogen removal only in nitrogen-sensitive coastal areas, even though nutrients can travel great distances to eutrophicate the Baltic Sea. The nitrogen removal capacity of wastewater treatment facilities can also fall short from time to time because of surges of very potent wastewater emanating from biogas plants.
"We have also participated in various Baltic Sea conservation projects, which have revealed that some biogas plants in the Baltic States, Eastern Europe and Belarus release untreated wastewater into waterways from where they flow downstream into the Baltic Sea," Mikola says unhappily.
Economic interests and growth in environmental awareness bode well for the method's export potential. Vahala notes that the emergence of a market for their product is also strongly dependent on legislation.
"If and when it emerges, we want to be prepared and biting at the bit to go ahead."
Provided by Aalto University