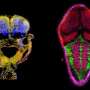
The role of tiny RNA in genetic diversity

All species, from zebrafish to humans, possess a genetically diverse collection of traits that allow them to adapt to changing environments. Yet scientists do not fully understand how organisms reach a state of optimal diversity—just enough variability to respond to environmental risks but not too much to function properly.
To explore the matter, a team of Yale researchers led by vascular biologist Stefania Nicoli studied zebrafish embryos, comparing traits of normal zebrafish to ones lacking specific RNA molecules. They found that the genetically modified embryos had greater range of traits or more diversity. For example, fish lacking a particular microRNA generate a population of sibling embryos with excessive blood vessel growth or a delay in growth. Such increased diversity made the mutant zebrafish more sensitive to environmental changes, such as oxygen deprivation, high-temperature stress, and toxic chemical exposure.
This is the first time researchers found that genetic diversity in a vertebrate animal is regulated by microRNA during very early development. Their diversity is inherited rather than acquired, Nicoli said. This finding, she noted, advances understanding of microRNA's role in development, and may in the future provide a tool to measure individual susceptibility to risk factors and diseases.
The study is published in Developmental Cell.
More information: Dionna M. Kasper et al. MicroRNAs Establish Uniform Traits during the Architecture of Vertebrate Embryos, Developmental Cell (2017). DOI: 10.1016/j.devcel.2017.02.021
Journal information: Developmental Cell
Provided by Yale University