Exploring the brain/fat connection to identify key differences amongst humans and monkeys
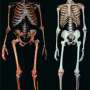
Among the greatest unanswered questions in all of evolutionary biology is what were the molecular forces that gave rise to the unique cognitive abilities of the human brain? Which, among the constellation of neurons, expanded amongst the myriad folds of gray and white matter in the cerebral cortex to vault humans over monkeys after the last common ancestor split some 6-8 million years ago?
In a tour-de-force comparative study, Professors Philipp Khaitovich, Patrick Giavalisco et al., utilized vast brain banks from across the world, to take a new approach to understanding uniquely human brain changes—the complete metabolic profile of fats, known as lipids, within the prefrontal cortex (PFC).
For the first time, they've shown that the lipid composition within the PFC is developmentally dynamic, and identified uniquely human changes in brain fats related to lifespan and composition. "We found that the greatest extent of lipid concentration changes takes place during the first several years of human life, which may be occurring as changes happen in the connectivity of the developing neocortex," said Philipp Khaitovich.
As a result, they have successfully identified and assemble the largest catalogue of lipid profile differences—more than 11,000 in total, amongst humans, chimpanzees and macaques.
In the new study, which appears in the advanced online edition of Molecular Biology and Evolution, the researchers wanted to develop a new approach to account for brain differences during development that are apparent in the course of a lifetime.
Since the majority of a brain's weight comes from fats and every brain axon signal is wrapped by an insulating fat sheath, called myelin, they thought that perhaps some of the mystery behind the human brain's higher functions, including learning, memory, sensory information—and ultimately individuality —may have a lipid connection. Furthermore, some brain lipid metabolism genes and proteins have been linked by genetic studies to human disorders including Alzheimer's (apoE4), Parkinson's, depression and anxiety.
To perform their fat composition analyses, the research team focused on a key brain area—the dorsolateral PFC across different ages, from 2 days to 61 years in humans, newborns to 42 years in chimpanzees, and 14 weeks to 21 years in macaques, with sampling skewed to early development. They took the samples from disease-free 40 individuals from each group, who died suddenly from unrelated causes.
They found more than 11,000 different fat compounds in their complete mass spectrometry analyses. More than two-thirds of the samples (7,589 lipid peaks) showed age-dependent concentration changes in humans, chimpanzees and macaques.
And in humans, the amount of change was greater during the first year of life than any year between 20 and 60 years of age. Within the human PFC, more than 61 percent of all lipid concentrations changes took place during the first 20 years of life. This same trend was found in chimpanzees and macaques.
They also found that the vital sheathing of axons from myelination specific lipids, especially cerebrosides, were enriched in the adult brain—-but myelin changes alone could not account for the majority of age-related changes.
Overall, the lipid changes are about 15 percent between infant and adult human PFC, with glycosphingolipids, triradylglycerols and glycosphingolipids showing the higher concentrations in adult brains, thought to be important in synapse formation and axonal myelination. In infant brains, ceramides, which are involved in cell signaling, differentiation and cell death, were in higher concentrations.
Next, they followed up the pre-frontal cortex lipidome measurements with a complete RNA sequencing, or transcriptome analysis, measuring levels of 13,777 mRNAs in 95 percent of all samples. They found a correlation between the age-dependent lipid changes and expression changes of enzymes, with 667 of the mRNAs associated with 2,064 age-dependent lipids within the human metabolome database.
An unexpected result from their studies is a new lipidome age test, based solely on the lipid profile of 27 age predictor lipids in humans, 24 in chimpanzees, and 19 in rhesus monkeys.
"We found that accuracy of age prediction to within a year-and-a half of their real age, and based solely on a lipid profile, is better than facial recognition," said Patrick Giavalisco.
Intriguingly, among species, humans showed greater lipid divergence compared to chimpanzees, with 251 lipids showing the most significant concentration differences between adult humans and chimpanzees, indicating relationship between emergence of cognitive skills unique to humans and brain's lipid composition.
Unexpectedly, most the greatest excess of human-specific differences was mapped not to early childhood, but to adulthood, from 20-35 years old in humans. These lipids functioned in breaking up fats within cells, vitamin digestion and absorption, fat digestion and insulin resistance.
Journal information: Molecular Biology and Evolution
Provided by Oxford University Press