New method detects residual contaminants in ultra-pure helium gas

The gas that makes balloons float is also vital to scientific experiments. In these experiments, natural helium (He) is purified, but it contains a tiny bit of a slightly different form of helium, known as the isotope 3He. A sample can contain just one 3He in every million helium atoms. That's too much for many experiments. Many experiments require ultra-pure helium, with a 3He component at least another million times smaller, or one in a trillion of the He atoms. Although techniques are believed to produce ultra-pure helium, until recently no experimental methods have confirmed that the amount of 3He present in a sample is indeed that small. Now, scientists at the ATLAS facility at Argonne National Laboratory have used accelerator mass spectrometry (AMS) to precisely measure the very small concentrations of 3He present.
Scientists need ultra-pure helium for a wide range of experiments. For example, they use ultra-pure helium to study the longevity and other properties of a free neutron. Free neutrons can provide insights into the formation of the universe and physics beyond the Standard Model, if measured accurately. To determine the purity of the helium for this study, the team demonstrated an approach that reaches a level of precision several orders of magnitude beyond that of any other technique. The team also found that measuring the amount of troublemaking 3He in purified helium samples intended for neutron studies suggest the need for significant experimental corrections, due to neutron absorption by the residual 3He present.
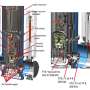
Answering difficult scientific questions about the nature of the universe requires isotopically purified helium (4He). The isotope 3He can contaminate the helium. Accurately measuring the amount of 3He requires determining the 3He/4He ratio at values well below those that can be achieved with standard mass spectroscopy techniques. Accelerator mass spectrometry provides the only way to directly measure the 3He content in purified helium samples at the level of sensitivity required for the neutron lifetime experiment, which seeks to determine how long a free neutron survives. Scientists used the ATLAS facility to demonstrate measurements of 3He/4He ratios as small as 10−14, or 1 in 100,000,000,000,000. In this work, scientists tuned the ATLAS accelerator, which serves as an ultra-precise mass filter, with specialized carbon ions. They scaled the accelerator components to 3He+. To reduce atmospheric 3He contamination, the team produced the 3He+ ions in a new radio frequency helium discharge source that reduced naturally occurring background sources of 3He. They monitored the final accelerator tune by regularly switching to H3+ ions from high-purity hydrogen. They eliminated H3+ ions and ions consisting of paired deuterium and hydrogen atoms by dissociation in a gold foil, after acceleration to 8 MeV. After stripping the second electron from the 3He+ ion, they dispersed the ions in a magnetic spectrograph and counted the 3He2+ ions. The team anticipates that these observations will also guide the design of future neutron experiments. Based on known improvements, an ultimate sensitivity to 3He/4He ratios as small as 10−15 appears to be feasible.
More information: H. P. Mumm et al. High-sensitivity measurement ofisotopic ratios for ultracold neutron experiments, Physical Review C (2016). DOI: 10.1103/PhysRevC.93.065502
Provided by US Department of Energy