Detecting potentially harmful mycotoxins in beer
Beer is one of the world's most popular alcoholic beverages. But, made with barley, brews can contain low levels of mycotoxins, which are produced by fungi that can contaminate grains. Although not a major health threat, the industry needs to minimize the risk of contamination. Now scientists have developed a portable sensor that can help. Their report appears in ACS' Journal of Agricultural and Food Chemistry.
Because of its alcohol content and the high temperatures required to make beer, most consumers might assume that contamination by biologically derived compounds is not an issue. But mycotoxins can survive the brewing process and end up in the final product. Some mycotoxins have been shown to cause genetic damage in cells and cancer in animals. Currently, methods to detect mycotoxin contamination in beer are costly and require in-laboratory analysis. Sweccha Joshi, Teris van Beek and colleagues wanted to come up with a less expensive, portable alternative.
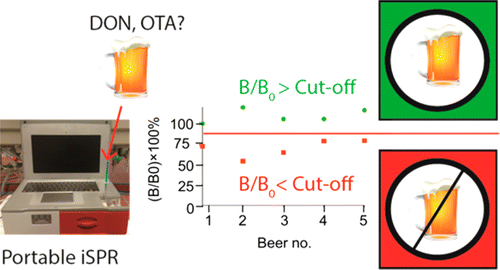
Building on technology used to detect mycotoxins in grains, the researchers developed a biosensing chip that can bind these compounds when they are present in beer samples. The team also could reuse the chip 450 times before it started to fail. Testing on commercial beer and barley showed that the portable instrument detected levels as low as 0.2 nanograms/milliliter of ochratoxin A and 120 ng/mL of deoxynivalenol—respectively, the estimated safe limits for these mycotoxins.
More information: Sweccha Joshi et al. Analysis of Mycotoxins in Beer Using a Portable Nanostructured Imaging Surface Plasmon Resonance Biosensor, Journal of Agricultural and Food Chemistry (2016). DOI: 10.1021/acs.jafc.6b04106
Abstract
A competitive inhibition immunoassay is described for the mycotoxins deoxynivalenol (DON) and ochratoxin A (OTA) in beer using a portable nanostructured imaging surface plasmon resonance (iSPR) biosensor, also referred to as imaging nanoplasmonics. The toxins were directly and covalently immobilized on a 3-dimensional carboxymethylated dextran (CMD) layer on a nanostructured iSPR chip. The assay is based on competition between the immobilized mycotoxins and free mycotoxins in the solution for binding to specific antibodies. The chip surface was regenerated after each cycle, and the combination of CMD and direct immobilization of toxins allowed the chips to be used for more than 450 cycles. The limits of detection (LODs) in beer were 17 ng/mL for DON and 7 ng/mL for OTA (or 0.09 ng/mL after 75 times enrichment). These LODs allowed detection of even less than 10% depletion of the tolerable daily intake of DON and OTA by beer. Significant cross-reactivity of anti-DON was observed toward DON-3-glucoside and 3-acetyl-DON, while no cross-reactivity was seen for 15-acetyl-DON. A preliminary in-house validation with 20 different batches of beer showed that both toxins can be detected at the considered theoretical safe level for beer. The assay can be used for in-field or at-line detection of DON in beer and also in barley without preconcentration, while OTA in beer requires an additional enrichment step, thus making the latter in its present form less suitable for field applications.
Journal information: Journal of Agricultural and Food Chemistry
Provided by American Chemical Society



















