September 15, 2016 report
Tracking technology shows some individual color-sensing cells in the eyes do not help us see color

(Phys.org)—A small team of researchers from the University of California and the University of Washington has found that not all of the color-sensing cells in the back of the eyeball actually help us to see color. Instead, as they note in their paper published in the journal Science Advances, some of them sense only white light.
A lot of research has gone into studying how the eye works, both in humans and in other animals, and researchers have learned much about the complicated anatomy that allows us to perceive the world in a rich array of colors and shapes. But until now, it has not been possible to study the activity of rods and cones (light sensing cells) in real-time in human volunteers. In this new effort, the researchers have overcome that challenge, and in so doing, have discovered something completely new about cone behavior.
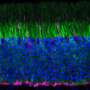
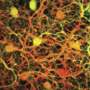
To gain a better understanding of the role that cones play in helping us interpret colors, the researchers asked two volunteers to sit still while their eyes were examined by an eye-tracking device based on the same telescope technology that allows astronomers to observe moving stars—called adaptive optics, it keeps the focus of a beam of light on a single point—in this case, on a single cone in the back of the eye. This allowed the researchers to conduct a survey of 273 cones for each volunteer, shining a single beam of light onto each cone and then asking the volunteer what they saw. In so doing, the researchers were surprised to find that approximately two-thirds of the cones did not register a sense of color at all, but instead perceived only white light.
Cones come in three varieties, L, M and S, each is able to sense different colors. But the researchers found that for all three types, 77 percent registered as white light sensing. They also found that the cells remained stable over time by testing the same cones over a period of many months—they suggest the purpose of having so many white sensing cells is to help define lines, edges and other details of objects in the world around us.
More information: R. Sabesan et al. The elementary representation of spatial and color vision in the human retina, Science Advances (2016). DOI: 10.1126/sciadv.1600797
Abstract
The retina is the most accessible element of the central nervous system for linking behavior to the activity of isolated neurons. We unraveled behavior at the elementary level of single input units—the visual sensation generated by stimulating individual long (L), middle (M), and short (S) wavelength–sensitive cones with light. Spectrally identified cones near the fovea of human observers were targeted with small spots of light, and the type, proportion, and repeatability of the elicited sensations were recorded. Two distinct populations of cones were observed: a smaller group predominantly associated with signaling chromatic sensations and a second, more numerous population linked to achromatic percepts. Red and green sensations were mainly driven by L- and M-cones, respectively, although both cone types elicited achromatic percepts. Sensations generated by cones were rarely stochastic; rather, they were consistent over many months and were dominated by one specific perceptual category. Cones lying in the midst of a pure spectrally opponent neighborhood, an arrangement purported to be most efficient in producing chromatic signals in downstream neurons, were no more likely to signal chromatic percepts. Overall, the results are consistent with the idea that the nervous system encodes high-resolution achromatic information and lower-resolution color signals in separate pathways that emerge as early as the first synapse. The lower proportion of cones eliciting color sensations may reflect a lack of evolutionary pressure for the chromatic system to be as fine-grained as the high-acuity achromatic system.
Journal information: Science Advances
© 2016 Phys.org