Botox's sweet tooth underlies its key neuron-targeting mechanism
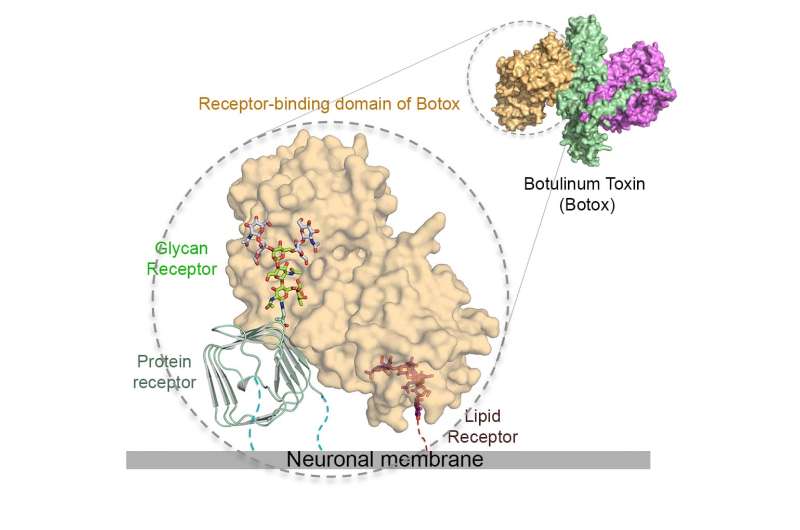
The Botox toxin has a sweet tooth, and it's this craving for sugars - glycans, to be exact - that underlies its extreme ability target neuron cells in the body ... while giving researchers an approach to neutralize it.
A study co-led by Rongsheng Jin, professor of physiology & biophysics at the University of California, Irvine; Min Dong with Boston Children's Hospital-Harvard Medical School; and Andreas Rummel with the Hannover Medical School in Germany, reveals an important general mechanism by which the pathogen is attracted to, adapts to and takes advantage of glycan modifications in surface receptors to invade motor neurons. Glycans are chains of sugars synthesized by cells for their development, growth, functioning or survival. Results appear June 13 in Nature Structural and Molecular Biology.
"Our findings reveal a new paradigm of the everlasting host-pathogen arms race, where a pathogen develops a smart strategy to achieve highly specific binding to a host receptor while also tolerating genetic changes on the receptor," Jin said. "And to some extent, this mechanism by which the toxin attacks human is similar to the one that is utilized by some important broad-neutralizing human antibodies to fight viruses, such as dengue viruses and HIV."
Botulinum neurotoxin A (BoNT/A), commonly known as the Botox toxin, is widely used in a weakened form for treating various medical conditions as well as for cosmetics. The clinical product Botox contains extremely low doses of the toxin and is safe to use. But at higher doses, it can be lethal, and it's also classified as a potential bioterrorism agent.
Its potency and therapeutic effects rely on its extraordinary ability to target motor neurons and subsequently block release neurotransmitters that control the movement of muscle fiber, which can result in paralysis. This specific targeting is achieved by highly selective interactions between the toxin and its receptors - host proteins that bind to the toxin in a lock and key manner.
The intricate detail of how the Botox toxin recognizes its receptors also reveals novel ways to neutralizing these deadly toxins. "With this new structural information," Rummel said, "we were able to pinpoint key amino acids in the toxin that are required for binding to sugars, and we found that even mutating a single amino acid is sufficient to abolish the toxicity by more than a million fold."
Remarkably, the newly mapped glycan-binding site turns out to be also the site recognized by a potent human neutralizing antibody called CR2/CR1, which is currently in clinical trial for the treatment and prevention of BoNT/A poisoning. "This antibody fights the toxin exactly by preventing it from binding to sugars on the receptor. Our study thus provides a new strategy for anti-toxin inhibitor design," Dong said.
Guorui Yao and Kwok-ho Lam at UCI, Sicai Zhang at Harvard, Stefan Mahrhold at the Hannover Medical School, Daniel Stern at Berlin's Center for Biological Threats & Special Pathogens, Kay Perry at the Argonne National Laboratory in Illinois, and Karine Bagramyan and Markus Kalkum the Beckman Research Institute at the City of Hope in Duarte, Calif., contributed to the study, which was primarily supported by the National Institutes of Health.
More information: N-linked glycosylation of SV2 is required for binding and uptake of botulinum neurotoxin A, Nature Structural and Molecular Biology, DOI: 10.1038/nsmb.3245
Journal information: Nature Structural and Molecular Biology
Provided by University of California, Irvine