Light sheet fluorescence microscope reveals key processes in the development of red flour beetles
A team of researchers at the University of Cologne has for the first time succeeded in observing the amniotic sac in insects. The red flour beetle (Tribolium castaneum), a pest of stored grains around the world, served as the object of investigation.
Using the example of the red flour beetle, the biologists are presenting a new model system for the exploration of the dynamic development of animal tissues. In this context, a high-tech light sheet fluorescence microscope was used for the first time on red flour beetles at the University of Cologne.
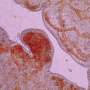
Early in development, a protective fluid-filled sac forms around an embryo. In humans, this sac bursts during birth, but the sac surrounding insect embryos ruptures long before these animals begin to emerge from their eggs. This early rupture is important for insects to develop normally: if an insect embryo's sac remains intact too long, the animal's back will not close properly.
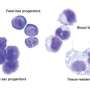
The epithelial tissue of the sac in the red flour beetle consists of two layers of tissue, the inner amnion and the outer serosa. However, in the past it was difficult to study what happens to the amnion as the insect embryo develops because it is hard to distinguish it from the serosa. Now, the research team has used genetically engineered red flour beetles in which the cells of the amnion produce a fluorescent protein that can be viewed under a microscope. This allowed the amnion to be observed in living specimens during beetle embryo development. With their observations, the researchers were able to disprove a model of the structure of these tissues that was developed eleven years ago. The biologists were now able to show that the two layers are discrete, but converge at various points in the animal's development. The results of the study are reported in eLife (dx.doi.org/10.7554/eLife.13834).
The interaction between the two tissue layers during the rupturing of the sac is particularly important for the insect's further development. Observing this process was made possible with a light sheet fluorescence microscope. The microscope's high resolution and speed allows the researchers to observe the breaking of the sac. "The small opening in the extraembryonic tissue then widens quickly to release the embryo," says Dr. Kristen Panfilio, the head of the Emmy Noether Independent Junior Research Group at the University of Cologne that conducted the research.
The researchers were further able to show that the cells in part of the amnion near the head of the beetle embryo have a special shape before the sac ruptures. "This region of the amnion breaks apart first, and the serosa breaks open a few minutes afterwards. Once the two layers have broken, together they pull back from the embryo like a pillowcase being turned inside out as it is removed from a pillow," Dr. Maarten Hilbrant from the research group explains. In normal beetles, this process is quite rapid and squeezes the embryo's abdomen. But in beetles genetically manipulated to lack a serosa, the process is slower because the amnion is not strong enough to squeeze the embryo by itself.
"The two tissues have to work together. The experiments show that the amnion starts the rupture of the red flour beetle embryo's protective sac and the serosa drives the process of the sac being peeled back," Panfilio concludes. Further research will now investigate the mechanics behind the two tissues' roles, and whether the amnion and serosa display similar behaviors in other related insect species.
The research was funded by the Emmy Noether Program of the German Research Foundation (DFG), with instrumentation support from the Collaborative Research Centers CRC 572 ("Commitment of Cell Arrays and Cell Type Specification") and CRC 680 ("Molecular Basis of Evolutionary Innovations"). Portions of the above text were taken from the eLife digest for this study, "The beetle amnion and serosa functionally interact as apposed epithelia."
More information: Maarten Hilbrant et al. The beetle amnion and serosa functionally interact as apposed epithelia, eLife (2016). DOI: 10.7554/eLife.13834
Journal information: eLife
Provided by University of Cologne