Molecular experiment reverses evolution in birds obtaining a dinosaur-like lower leg
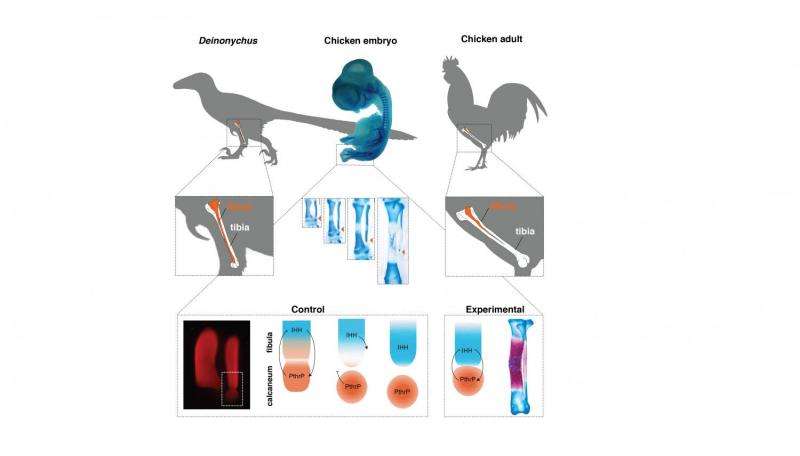
Anyone who has eaten roasted chicken can account for the presence of a long, spine-like bone in the drumstick. This is actually the fibula, one of the two long bones of the lower leg (the outer one). In dinosaurs, the ancestors of birds, this bone is tube-shaped and reaches all the way down to the ankle. However, in the evolution from dinosaurs to birds, it lost its lower end, and no longer connects to the ankle, being shorter than the other bone in the lower leg, the tibia. Scientists noted long ago that bird embryos first develop a tubular, dinosaur-like fibula. Afterward, it becomes shorter than the tibia and acquires its adult, splinter-like shape.
Brazilian researcher Joâo Botelho, working at the lab of Alexander Vargas (University of Chile) studied the mechanisms that underlie this transformation. In normal bone development, the shaft matures and ceases growth (cell division) long before the ends do. Botelho found that molecular mechanisms of maturation were active very early at the lower end, ceasing cell division and growth. Inhibiting a maturation gene called Indian Hedgehog resulted in chickens with a tubular fibula as long as the tibia and connected to the ankle, just like a dinosaur.
Botelho and collaborators believe that early maturation at the lower end of the fibula occurs because of the influence of a nearby bone in the ankle, the calcaneum. Unlike other animals, the calcaneum in bird embryos presses against the lower end of the fibula: They are so close they have even been mistaken for a single element by some researchers. Botelho proposes that at this stage, the lower end of the fibula receives signals more like those at the bone shaft. In normal development, the calcaneum then becomes detached from the fibula. However, its distal end has already become committed to shaft-like development, and matures early. In the chickens with experimentally dinosaur-like lower legs, the calcaneum was attached to the fibula. Botelho also confirmed the calcaneum strongly expresses PthrP, a gene that allows growth at the ends of bones.
Another interesting observation in the experimental chickens was that the other bone of the lower leg, the tibia, was significantly shorter. This suggests that a dinosaur-like fibula connected to the ankle stops the tibia from outgrowing the fibula, as it normally would. Working with Jingmai O'Connor (IVPP, China), the research team realized this was consistent with an evolutionary pattern documented by the fossil record. The earliest forms to evolve reduced fibulas were toothed birds from the early cretaceous age, which lived alongside dinosaurs. These forms had splinter-like fibulas that did not connect to the ankle, but were almost as long as the tibia. The fibula first lost its lower end in evolution. This may have allowed the evolution of tibias that are much longer than the fibula, which occurred afterwards.
The results of the entire study have been published this week in the renowned academic journal Evolution . This is the second time Botelho has achieved an experimental reversal to a dinosaur-like trait in birds. Previously, he had managed to undo the evolution of the perching toe of birds to produce a non-twisted, non-opposed toe, as in dinosaurs. Another lab at Yale obtained a dinosaur-like snout by altering gene expression in embryonic chicken. However, these studies are not aimed at producing dinosaurs for commercial or non-scientific purposes, as in the Jurassic Park series.
"The experiments are focused on single traits to test specific hypotheses," says Vargas. "Not only do we know a great deal about bird development, but also about the dinosaur-bird transition, which is well-documented by the fossil record. This leads naturally to hypotheses on the evolution of development, that can be explored in the lab."
More information: João Francisco Botelho et al. Molecular development of fibular reduction in birds and its evolution from dinosaurs, Evolution (2016). DOI: 10.1111/evo.12882
João Francisco Botelho et al. Skeletal plasticity in response to embryonic muscular activity underlies the development and evolution of the perching digit of birds, Scientific Reports (2015). DOI: 10.1038/srep09840
Bhart-Anjan S. Bhullar et al. A molecular mechanism for the origin of a key evolutionary innovation, the bird beak and palate, revealed by an integrative approach to major transitions in vertebrate history, Evolution (2015). DOI: 10.1111/evo.12684
Journal information: Evolution , Scientific Reports
Provided by Universidad de Chile