Restoring the American chestnut

The American chestnut was once a mainstay in hardwood forests as far north as Maine and as far south as Georgia and Mississippi. A massive chestnut blight in the early part of the 20th century ended the mighty chestnut's domination, wiping out billions of mature trees. Scientists are now working to restore the American chestnut's place in U.S. forests. A study published in the February issue of HortScience provides new recommendations that can help increase the stock of blight-resistant trees.
According to the authors, genetic engineering is now poised to offer a solution: blight-resistant American chestnut trees. "The addition of one gene, oxalate oxidase, can protect the American chestnut by breaking down the oxalic acid secreted by the fungus," explained Dr. Allison Oakes, corresponding author of the study. The fungus still infects wounds in the bark, but after the initial infection the resistant tree halts the spread of the fungus behind a layer of plant tissue.
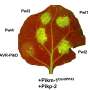
Oakes and colleagues Tyler Desmarais, William Powell, and Charles Maynard from the State University of New York College of Environmental Science and Forestry say that hundreds of American chestnut trees are needed for field trials and eventual restoration plantings, but noted that "production is bottlenecked" because of the difficulty of making hardwood trees produce roots through micropropagation. "The presence of roots and living shoot tips precede successful acclimatization of tissue culture-produced American chestnut plantlets," they explained. To improve the post-rooting stage of American chestnut propagation protocol, the scientists designed four experiments in which they examined vessel type, concentrations of humic acid and activated charcoal, and use of a vermiculite substrate.
Results showed that the presence of activated charcoal in the postrooting medium significantly increased the percentage of rooted plantlets, but increasing the concentration did not significant affect root presence. However, increasing concentrations of activated charcoal did have a significant effect on number of roots, and length of the longest root.
In regard to vessel type, results showed that using disposable clamshell containers during postrooting significantly decreased the survival of shoot tips, but significantly increased root formation. The presence of activated charcoal across vermiculite treatments increased root presence and number of roots while decreasing shoot tip survival. "High concentrations of humic acid combined with activated charcoal had excellent and significantly higher root presence than all the treatments without activated charcoal," the authors noted. They added that this trend was similar for root number.
According to the report, an "overarching trend" evidenced across the experiments was that activated charcoal enhanced root growth but slightly decreased shoot tip survival.
"Our findings have broad implications for tissue culture and genetic engineering of target hardwood species," Oakes said. "The combination of humic acid and activated charcoal in rooting medium more than doubled the number of roots produced by micropropagated shoots than either compound alone. Vermiculite substrate may be an alternative with additional research and methods development."
The authors said that the recommendations could be applicable to other difficult-to-root hardwood trees in transgenic programs, such as American butternut, white oak, and black walnut.
More information: HortScience, hortsci.ashspublications.org/c … nt/51/2/171.abstract
Journal information: HortScience
Provided by American Society for Horticultural Science