February 10, 2016 report
Improved nuclear magnetic resonance technique allows researchers to measure signals from a single molecule

(Phys.org)—An effort by a combined team of researchers from Harvard and Ulm Universities has led to the development of an improved nuclear magnetic resonance technique that allows for better measuring the magnetic field of a single molecule. In their paper published in the journal Science, the team describes the new technique and the results of experiments using it to detect molecules of a specific protein.
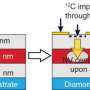
Scientists have used a technique called nuclear magnetic resonance (NMR) spectroscopy for quite some time to learn more about the structure of molecules—originally it involved using radio waves to measure atomic nuclei lining up due to a strong magnetic field. The technique was later improved by using the magnetic properties of a single nitrogen vacancy (NV) in a diamond to measure the molecular fingerprints that became evident when magnetic moments coupled to one another. A NV in a diamond comes to exist when instead of a carbon atom inside the diamond, a defect occurs that results in a vacancy and a nitrogen atom. In this new effort the researchers found two ways to improve on this technique to allow for improved coherence.
The first improvement involved moving the NV sensor closer to the diamond in a way that did not cause quantum coherence to be degraded by outside noise. The second improvement involved creating a new read-out procedure that allowed the team to measure the magnetic moment of the NV center without causing it to change, by employing quantum entanglement with the nitrogen atom's nuclear magnetic moment. Taken together the changes allowed for a type of NMR spectroscopy that was 500 times as sensitive as conventional techniques.
The researchers then used the technique to take measurements of a protein called ubiquitin by placing a molecule sample onto the surface of the diamond and then detecting properties of individual molecules—in this case, they tested and report that they were able to detect carbon-13 and spectra of hydrogen-2 nuclei, when both were enriched with isotopes.
The team plans to continue working on their technique, hoping to find ways to improve it further—one approach they plan to study is whether location placement on the diamond makes any difference.
More information: I. Lovchinsky et al. Nuclear magnetic resonance detection and spectroscopy of single proteins using quantum logic, Science (2016). DOI: 10.1126/science.aad8022
Abstract
Nuclear magnetic resonance (NMR) spectroscopy is a powerful tool for the structural analysis of organic compounds and biomolecules, but typically requires macroscopic sample quantities. We utilize a sensor, consisting of two quantum bits corresponding to an electronic spin and an ancillary nuclear spin, to demonstrate room temperature magnetic resonance detection and spectroscopy of multiple nuclear species within individual ubiquitin proteins attached to the diamond surface. Using quantum logic to improve readout fidelity and a surface treatment technique to extend the spin coherence time of shallow NV centers, we demonstrate magnetic field sensitivity sufficient to detect individual proton spins within one second of integration. This gain in sensitivity enables high-confidence detection of individual proteins and allows us to observe spectral features that reveal information about their chemical composition.
Journal information: Science
© 2016 Phys.org