Making liver tissue in the lab for transplants and drug screening

Engineered liver tissue could have a range of important uses, from transplants in patients suffering from the organ's failure to pharmaceutical testing. Now scientists report in ACS' journal Analytical Chemistry the development of such a tissue, which closely mimics the liver's complicated microstructure and function more effectively than existing models.
The liver serves a critical role in digesting food and detoxifying the body. But due to a variety of factors, including viral infections, alcoholism and drug reactions, the organ can develop chronic or acute problems. When it doesn't work well, a person can suffer abdominal pain, swelling, nausea and other symptoms. Complete liver failure can be life-threatening and can require a transplant, a procedure that currently depends on human donors. To curtail this reliance and provide an improved model for predicting drugs' side effects, scientists have been engineering liver tissue in the lab. But so far, they haven't achieved the complex architecture of the real thing. Jinyi Wang and colleagues came up with a new approach.
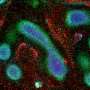
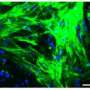
Wang's team built a microfluidics-based tissue that copies the liver's complex lobules, the organ's tiny structures that resemble wheels with spokes. They did this with human cells from a liver and an aorta, the body's main artery. In the lab, the engineered tissue had a metabolic rate that was closer to real-life levels than other liver models, and it successfully simulated how a real liver would react to various drug combinations. The researchers conclude their approach could lead to the development of functional liver tissue for clinical applications and screening drugs for side effects and potentially harmful interactions.
More information: Chao Ma et al. On-Chip Construction of Liver Lobule-like Microtissue and Its Application for Adverse Drug Reaction Assay, Analytical Chemistry (2016). DOI: 10.1021/acs.analchem.5b03869
Abstract
Engineering the liver in vitro is promising to provide functional replacement for patients with liver failure, or tissue models for drug metabolism and toxicity analysis. In this study, we describe a microfluidics-based biomimetic approach for the fabrication of an in vitro 3D liver lobule-like microtissue composed of a radially patterned hepatic cord-like network and an intrinsic hepatic sinusoid-like network. The hepatic enzyme assay showed that the 3D biomimetic microtissue maintained high basal CYP-1A1/2 and UGT activities, responded dynamically to enzyme induction/inhibition, and preserved great hepatic capacity of drug metabolism. Using the established biomimetic microtissue, the potential adverse drug reactions that induced liver injury were successfully analyzed via drug–drug interactions of clinical pharmaceuticals. The results showed that predosed pharmaceuticals which agitated CYP-1A1/2 and/or UGT activities would alter the toxic effect of the subsequently administrated drug. All the results validated the utility of the established biomimetic microtissue in toxicological studies in vitro. Also, we anticipate the microfluidics-based bioengineering strategy would benefit liver tissue engineering and liver physiology/pathophysiology studies, as well as in vitro assessment of drug-induced hepatotoxicity.
Journal information: Analytical Chemistry
Provided by American Chemical Society