How close are we to successfully editing genes in human embryos?
An important international summit on human gene editing recently recommended that researchers go ahead with gene editing human embryos, but keep revisiting how and when such modifications would be appropriate in the clinic. The decision came after some scientists called for a moratorium on such research.
The recommendation was always going to be controversial, with many people concerned that the technology, which could be used to prevent parents from passing on genetic diseases to their children, will be misused and lead to permanent changes in the human gene pool.
But how close are we – is there really reason to be concerned at this point?
Laboratory promise
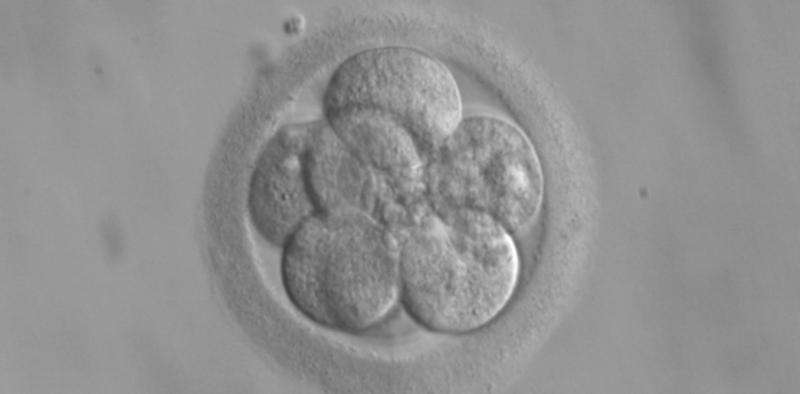
Gene editing of the human germline – those cells that form the sperm and eggs and, from a fertilised egg, will generate every cell in the human body – is different from other types of genetic editing because changes in those cells will be inherited by future generations, to become a permanent change in the human make-up.
Working on human germline cells at the very earliest stages of the formation of an embryo, just after an egg has been fertilised and then implants itself in the womb, is of course impossible to do in a pregnant woman. In my lab, where our focus is on early development, we approach this research using mice and, more recently, by simply growing human cells in a culture dish. In this way we have managed to identify some of the earliest genetic events that "specify" a stem cell to become a germline cell.
At the same time the technology underpinning gene editing, such as the CRISPR/Cas9 – a fast, easy and unprecedentedly precise method for targeting edits to specific genes – is becoming widespread across science. Together with the new ways of studying germline cells in the lab, this is offering a real chance for scientists and the public to consider whether or not editing of the human germline has merit – before any harm can be done.
We can now create human "primordial germ cells", the precursors to eggs and sperm, from embryonic stem cells. It is a delicate and time-consuming procedure, and the resulting cells do not survive beyond the very early stages of development – partly because we have yet to reproduce the conditions that they are designed to thrive in. What we have been able to show is that some of the earliest steps in the development of human primordial germ cells are different from those in mice. This is important as most of the previous results in this area have come from mouse models, indicating that such information cannot actually be wholly extrapolated to describe humans.
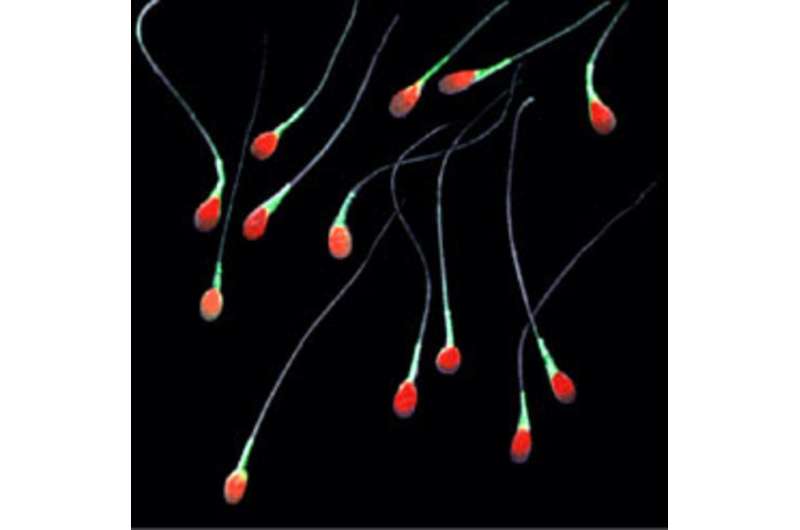
Last year, we also managed to generate primordial germ cells from adult body cells, such as human skin cells. We take body cells that have been programmed to revert back into stem cells, and add chemical factors to "re-specify" them as primordial germ cells.
While these cells don't survive long either, experiments have shown that introducing such cells into the testes and ovaries of in mice does allow them to continue their development and maturation into sperm and eggs. Remarkably, such mice were able to give birth to healthy offspring raising the prospect of reprogrammed skin cells creating living human beings. For that reason it certainly makes sense to carry out similar studies using primates. Further research might also make it possible to develop working sperm and egg cells entirely in a culture dish.
Finished blueprint?
Looking ahead, it is clear that there already is a potential template for editing the human germline. Genome-sequencing methods could also provide for additional checks to ensure that no inadvertent mutations or "off-target" effects have occurred during the editing procedures.
What's more, if viable sperm and eggs could be grown in the lab from primordial germ cells, they could be used to generate fertilised embryos. Such "pre-implantation" embryos could also be further screened (as is routine now in the in-vitro fertilisation procedure) to ensure transfer to the womb of only those embryos that are free from specific mutations.
So how could this work in a clinic? Imagine combining the procedures in one patient, for example a woman with a disease-causing mutation who does not wish to pass this mutation to her child. Starting with a cell taken from her skin, this is reprogrammed to a primordial germ cell, in which the DNA is then edited to remove the mutated gene. The primordial germ cell is developed into an egg and used to create an embryo for IVF, to be screened and transplanted back into her womb. The child and its subsequent descendants would be free of the mutated gene.
There's a reason why the summit carefully considered such massive implications and nevertheless recommended to pursue such research. Without making further gains in our knowledge about the fundamental processes in early germ cell and embryo development – starting with growing germ cells for longer in the culture dish – we will not know what we can and cannot safely achieve with the new gene-editing technologies. We are still some way from being able to contribute the necessary biological evidence to society's debate about which, if any, of these technologies to pursue.
Source: The Conversation
This story is published courtesy of The Conversation (under Creative Commons-Attribution/No derivatives).
![]()