Breakthrough allows tracking of single molecules in 3-D with nanoscale accuracy
An innovative approach to calibrating high-tech microscopes enables researchers to track the movement of single molecules in 3D at the nanoscale.
A Stanford University research team, led by W. E. Moerner, extends the work that earned Moerner and colleagues Eric Betzig and Stefan W. Hell the 2014 Nobel Prize for Chemistry. Betzig and Moerner pioneered the development of super-resolution imaging, which broke the diffraction limit of optical microscopy by using the fluorescence of single molecules for the first time. The new work, published in The Optical Society's high impact journal Optica, demonstrates a marked improvement in the accuracy of this imaging technique and for tracking molecules in three dimensions.
Tracking how molecules move, form shapes and interact within the body's cells and neurons offers a powerful new view of key biological processes such as signaling, cell division and neuron communication, all of which impact people's health and susceptibility to disease.
Capitalizing on a Transformation in Microscopy
Super-resolution microscopy uses lasers to excite fluorescence from single molecules under conditions where only a few are emitting at a time, overcoming the traditional resolution limit for optical microscopy set by the diffraction limit of light.
"With the advent of super-resolution imaging, we improved the resolution by a factor of 5 to 10 beyond the diffraction limit – from 200 nanometers down to 40 or even 10 nanometers," Moerner said. "This new world of greatly increased resolution brings a big transformation in how the optical system works."
However, previous calibration techniques for super-resolution microscopy were not sufficiently accurate for 3D measurements of single molecules. The new calibration method uses a nanohole array to correct for optical distortions across a widefield microscope's entire field of view.
Dealing with Distortion
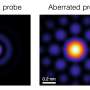
When imaging at the scale of single molecules, a single point of light coming from a molecule can typically be located with around 10-nanometer precision. At such high resolutions, any small imperfections in an optical system introduce image distortions, or aberrations, which can significantly skew measurements, particularly in 3D. The resulting errors could mean the difference between interpreting two molecules as interacting or simply being close to each other.
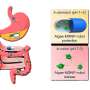
While many use fluorescent beads to calibrate 3D microscopes, Alex von Diezmann, doctoral candidate at the Moerner Lab, Stanford University, took a different approach. He created an array of holes in a metal film, each smaller than 200 nanometers and regularly spaced 2.5 microns apart, to use as a 3D calibration standard. Once the holes were filled with fluorescent dyes, the array could be used to calibrate for optical errors across the microscope's entire field of view, not just at a few select spots, as is possible using fluorescent beads. Using this technique, the researchers were able to correct aberrations of 50-100 nanometers to just 25 nanometers.
"Prior to this, people had not explicitly worried about these aberrations," von Diezmann said. "The fact that we demonstrated the presence of field-dependent aberrations, and showed that they could degrade images, is an important part of this work."
The researchers studied the new calibration technique with double-helix and astigmatic point spread functions, two types of optical modification typically used to extract z-axis location. Although both point spread functions showed z-axis related inaccuracies that created about a 20 percent error in the 3D measurements, the researchers corrected these aberrations using the 3D nanohole array.
Demonstrating Benefits for Study of Proteins in Bacteria
The researchers are now applying the new 3D calibration technique to all their single-molecule tracking and super-resolution microscopy studies. For example, von Diezmann is using it to study protein localization in bacteria that measure only two microns in length. With the 3D calibration technique, he can accurately measure and track key signaling proteins in nanodomains that are only 150 to 200 nanometers in size.
The researchers point out that correcting field-dependent and other types of aberrations is becoming more and more important as optical microscopy techniques evolve to image deeper into cells, for example.
"We studied this approach for a couple of cases, but it can be used with any super-resolution or localization microscopy that requires really precise 3D measurements," said von Diezmann. "It will be exciting to see other groups use it to figure out how their particular technique is affected by field-dependent aberrations. As a community, maybe we can find even better ways of dealing with these aberrations."
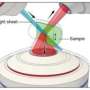
Researchers produced a 3D calibration tool by creating an array of nanoscale holes filled with fluorescent dye. In (a), widefield illumination (green) passes through the glass coverslip into a nanohole etched into a layer of aluminum. The solution of fluorescent dye fills the holes, and the resulting points of light (orange) are detected from below. Figure (b) shows a scanning electron microscope image of the holes, which are each 200 nanometers or less in diameter.
More information: Alex von Diezmann et al. Correcting field-dependent aberrations with nanoscale accuracy in three-dimensional single-molecule localization microscopy, Optica (2015). DOI: 10.1364/OPTICA.2.000985
Journal information: Optica
Provided by Optical Society of America