Researchers find out why high-performance glass flows, and how fast
Does glass in cathedral windows flow downward at room temperature—acting essentially as a liquid in super slow-motion? No. It's a myth. The larger volume observed at the bottom of some windows is due to the manufacturing process, not gravity.
But in more complex, high-tech kinds of glass, like Corning Gorilla Glass, a scratch- and damage-resistant glass used on more than one billion smartphones and tablets screens, susceptibility to room-temperature deformations have been known to exist for a few years. Now researchers from the UCLA Henry Samueli School of Engineering and Applied Science have discovered why such flowing happens and how fast.
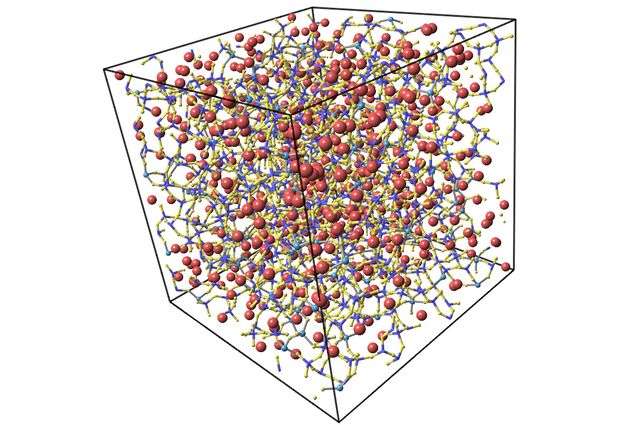
Using molecular dynamics simulations of different glasses, the researchers showed that high-performance glass can exhibit some long-term deformations that are proportional to how large the piece of glass is. Glass on your mobile phone's screen might not flow at all in its short lifetime, but large screens, such as those for giant TV screens can start to flow during the first few months after the glass is manufactured at a tiny but perceptible rate, about 10 micrometers per year for a one-meter-square piece of glass. While rate may seem slow, the effect is enough to limit how large these screens can be, the researchers said.
The research, led by Mathieu Bauchy, assistant professor of civil and environmental engineering, was reported today in the journal Physical Review Letters.
"We found that this long-term relaxation is due to the coexistence of competitive chemical elements of different sizes in the atomic network of the glass, which is known as the mixed-alkali effect," Bauchy said.
The key is the use of two alkali ions, sodium and potassium, in the high-performance glass formation process. Sodium and potassium ions are typically added into the composition of glasses as their presence lowers the temperature needed to form glass, thereby saving energy. However, this process and combination of ions with different atomic sizes makes glass susceptible to long-term deformations, the researchers found.
"Although the origin of the mixed-alkali effect remains debated, there are strong evidences that the size mismatch between the alkali atoms and the surrounding atomic network acts as a driving force to stimulate relaxation," Bauchy said. "In a sense, any alkali ion that is not satisfied with the size of its local pocket will want to jump to another one that better fits."
This phenomenon is also considered the source of the "thermometer effect." In the 19th century, most thermometers were made of glass containing equal portions of sodium and potassium, and revealed to gradually become inaccurate because of the deformation of the glass.
"The next step will be to prescribe optimal glass compositions that feature little, if any, relaxation, in order to enable the design of large yet stable screens," he said.
More information: "Stretched Exponential Relaxation of Glasses at Low Temperature." Phys. Rev. Lett. 115, 165901 – Published 15 October 2015. dx.doi.org/10.1103/PhysRevLett.115.165901
Journal information: Physical Review Letters
Provided by University of California, Los Angeles