Team studies light-induced oxidation of two-dimensional black phosphorus
University of Arkansas researchers, in collaboration with an international team, discovered that the energy required for oxidation of black phosphorus is reduced around surface defects, opening the door for further research into how to prevent oxidation in the future.
Their paper, "Intrinsic defects, fluctuations of the local shape, and the photo-oxidation of black phosphorus," was published by the American Chemical Society and featured this week at ACS Central Science's main page.
University of Arkansas researchers Kainen L. Utt, Pablo Rivero, Mehrshad Mehboudi and Salvador Barraza-Lopez of the Department of Physics and Edmund O. Harriss of the Department of Mathematical Sciences collaborated on the project with Mario F. Borunda, Department of Physics, Oklahoma State University, and Alejandro A. Pacheco SanJuan, Department of Mechanical Engineering, Universidad del Norte, Colombia.
"We have shown in this research how critical defects are in understanding the material's degradation," Barraza-Lopez said. "This opens the door to look at how to use chemical routes to prevent degradation of black phosphorus."
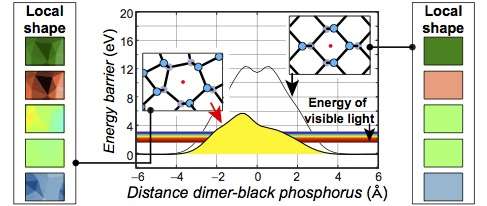
Black phosphorus degrades when exposed to oxygen. But the team found that the energy barrier that enables such degradation is within the range of visible light around imperfections of the black phosphorus. This means that the material readily oxidizes at these defects upon exposure to light.
Furthermore, oxygen dimers became split into single atoms at the end of the oxidation process, in a process called "dissociation." This study thus provides the first plausible mechanism as to how such observed dissociated oxygen dimers can be created. They further document an increase of the curvature of the material upon oxidation.
"This work would not have been possible without computational support from the High-Performance Computing Center at Arkansas and NSF-XSEDE," said Barraza-Lopez.
More information: "Intrinsic Defects, Fluctuations of the Local Shape, and the Photo-Oxidation of Black Phosphorus" ACS Cent. Sci., 2015, 1 (6), pp 320–327 DOI: 10.1021/acscentsci.5b00244
Provided by University of Arkansas




















