Tackling the root cause of cystic fibrosis
Treatments for cystic fibrosis (CF) have added years to the lives of thousands of Americans. But they can be difficult to administer, and most don't fix the underlying cause. Scientists have now found that a small molecule, when tested in yeast, can substitute for a protein and restore a key cellular function related to those missing in people with CF and similar conditions. Their report appears in the Journal of the American Chemical Society.
CF is caused by a genetic mutation that affects certain cellular proteins that make up "channels," which act like gates managing the flow of ions in and out of cells. In CF, some of these gates don't work like they're supposed to. This ultimately leads to a thick buildup of mucus in the lungs and other organs that can make it difficult for people with the disease to breathe. Treatment can involve inhaled medicines, enzyme supplements at every meal and clearing out airways, which sometimes requires the help of others. Martin Burke and colleagues wanted to find a way to better address the root cause of the disease.
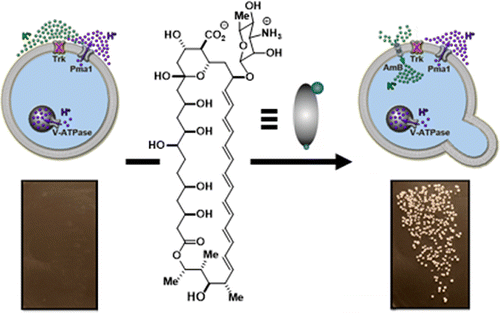
The researchers turned to a natural small molecule called amphotericin B, which was originally extracted from bacteria and is used to treat fungal infections. They tested it at low doses on a strain of yeast that is unable to transport ions in and out of its cells, and therefore doesn't grow. But with treatment, the yeast grew nearly as well as the control. The researchers say this shows that channel-forming small molecules can work in collaboration with protein pumps and channels in cell membranes to restore the flow of ions. They also suggest that while it remains to be seen whether this approach can be effective in human diseases like cystic fibrosis, the results in yeast provide a mechanistic framework for pursuing such a therapeutic strategy.
More information: Restored Physiology in Protein-Deficient Yeast by a Small Molecule Channel, J. Am. Chem. Soc., 2015, 137 (32), pp 10096–10099. DOI: 10.1021/jacs.5b05765
Abstract
Deficiencies of protein ion channels underlie many currently incurable human diseases. Robust networks of pumps and channels are usually responsible for the directional movement of specific ions in organisms ranging from microbes to humans. We thus questioned whether minimally selective small molecule mimics of missing protein channels might be capable of collaborating with the corresponding protein ion pumps to restore physiology. Here we report vigorous and sustainable restoration of yeast cell growth by replacing missing protein ion transporters with imperfect small molecule mimics. We further provide evidence that this tolerance for imperfect mimicry is attributable to collaboration between the channel-forming small molecule and protein ion pumps. These results illuminate a mechanistic framework for pursuing small molecule replacements for deficient protein ion channels that underlie a range of challenging human diseases.
Journal information: Journal of the American Chemical Society
Provided by American Chemical Society